Correct option is C
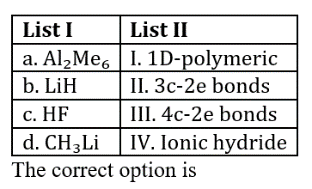
A three-center two-electron (3c–2e) bond is an electron-deficient chemical bond where three atoms share two electrons. The combination of three atomic orbitals form three molecular orbitals: one bonding, one non-bonding, and one anti-bonding. The two electrons go into the bonding orbital, resulting in a net bonding effect and constituting a chemical bond among all three atoms. In many common bonds of this type, the bonding orbital is shifted towards two of the three atoms instead of being spread equally among all three. This bonding pattern is seen in trimethylaluminium, which forms a dimer Al2(CH3)6 with the carbon atoms of two of the methyl groups in bridging positions. In Al2(CH3)6, there are two such 3c-2e bonds: two CH3 groups bridge the two Al atoms, leaving two additional CH3 groups in ordinary Al-CH3 bonds on each Al. As a result, the molecule achieves stability since each Al participates in a total of four bonds and all bonding molecular orbitals are filled, although two of the four bonds are 3-center Al−CH3−Al bonds.
Lithium hydride is an inorganic compound with the formula LiH.
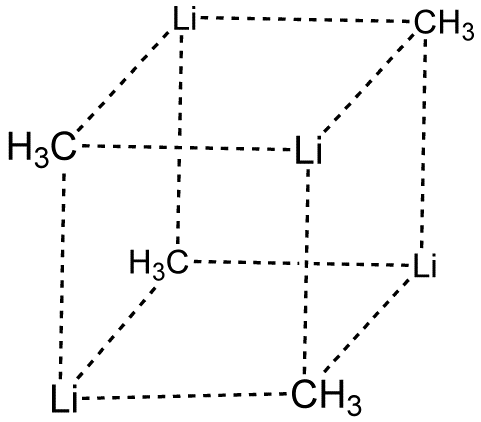
Methyllithium is the simplest organolithium reagent, with the empirical formula CH3Li.This s-block organometallic compound adopts an oligomeric structure both in solution and in the solid state.
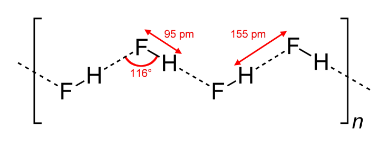
HF is diatomic in the gas-phase. As a liquid, HF forms relatively strong hydrogen bonds, hence its relatively high boiling point. Solid HF consists of zig-zag chains of HF molecules. The HF molecules, with a short covalent H–F bond of 95 pm length, are linked to neighboring molecules by intermolecular H–F distances of 155 pm. Liquid HF also consists of chains of HF molecules, but the chains are shorter, consisting on average of only five or six molecules.