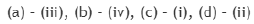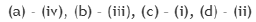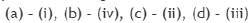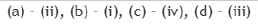- UGC NEW MEGAPACKS
- UGC NET JUNE
- UGC Crash Course
- UGC NET English
- UGC NET Commerce
- UGC NET Home Science
- UGC NET Management
- UGC NET Education
- UGC NET Pol Science
- UGC NET History
- UGC NET Hindi
- UGC NET EVS
- UGC NET LAW
- UGC NET Physical Education
- UGC NET Economics
- UGC NET Geography
- UGC NET Computer Science
- UGC NET Psychology
- UGC NET Sociology
- UGC NET Music
- UGC NET December
- UGC NET Sanskrit
- UGC NET Urdu
- UGC NET Philosophy
- UGC NET SOCIAL WORK
- UGC NET VISUAL ARTS
- JRF PREP
- RPSC Assistant Professor
- MP Assistant Professor
- UGC NET Video Course
- UGC NET Dual Edge
- UGC NET Ayurveda Biology
- UGC Exams Books Kit
- Ph.D
- Chhattisgarh SET
- Haryana Assistant Professor
- UGC WORKSHOP
- BIHAR ELIGIBILITY TEST
- AI Skills
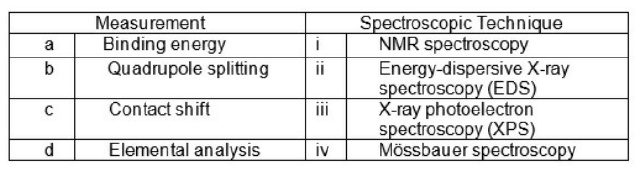
Match the following:
Similar Questions
- 1)
Mossbauer spectrum of at liquid nitrogen temperature in the presence of a field gradient and taking into effect quadrupole interaction [For 57Fe,I=1/2 for the ground state and I = 3/2 for the first excited state], shows a
- 2)
Consider the 31P NMR (decoupled from 1H and 195Pt) spectra for the isomers of 'A' and 'B' [IrCl3(PMe3)3], separately. The number of 31P NMR signals for the different isomers of the complexes are given in the table below. The correct answer is set.
- 3)
Among the following statements, select those which best describe plastocyanine
A. Coordination geometry of Cu2+\text{Cu}^{2+}CuIICu^{II}CuII
-ion is distorted tetrahedron
B. The geometry around Cu2+\text{Cu}^{2+}
changes to square-planar on reduction
C. In EPR spectrum, the A∥ values are smaller in plastocyanine than in
D. The active site contains the amino acids histidine and cysteine onlyChoose the correct answer from the options given below:
- 4)
Th ground state term symbol and the value for ion, respectively are (Atomic number of Pr is 59)
- 5)
Consider the following reactions.
The correct stereochemical dispositions of the Br ligands in A and the CO ligands in B, respectively, are
- 6)
In solution, Vaska's complex is known to bind reversibly. In the product, oxidation state of the metal ion center coordination geometry and mode of binding of , respectively are
- 7)
The reaction(s)/ event(s) used to prove the existence of neutrinos is/are
A.
B.
C.
Choose the correct answer from the options given below:
- 8)
Isolobal pairs from the fragments (A-D) is/are
A. {BH}
B.
C. {P}
D.
Choose the correct answer from the options given below:
- 9)
Among the following feasible reaction(s) is/are?
A.
B.
C.
Choose the correct answer from the options given below:
- 10)
Consider following statements regarding the magnetic moment of the 3d transition metal complexes:
A. Octahedral complexes of (high spin) and (high spin) have approximately equal to their values.
B. Tetrahedral complexes of and octahedral complexes of have orbital contribution added to their magnetic moments.
C. Orbital contribution is quenched for metal ions having A or E ground state.
D. Octahedral complexes of have value at 300 K due to orbital contribution added to their magnetic moments
Choose the correct answer from the options given below:

Access ‘CSIR NET Chemical Sciences’ Mock Tests with
- 60000+ Mocks and Previous Year Papers
- Unlimited Re-Attempts
- Personalised Report Card
- 500% Refund on Final Selection
- Largest Community

Access ‘CSIR NET Chemical Sciences’ Mock Tests with
- 60000+ Mocks and Previous Year Papers
- Unlimited Re-Attempts
- Personalised Report Card
- 500% Refund on Final Selection
- Largest Community