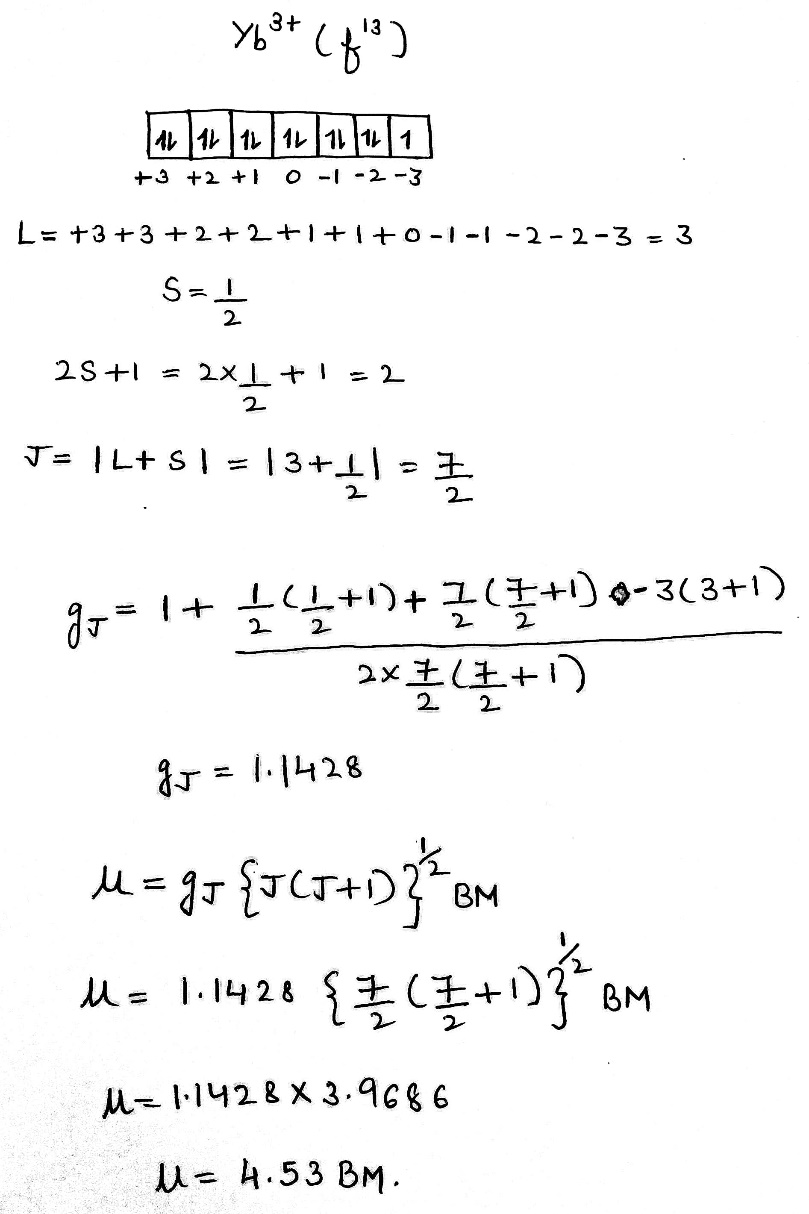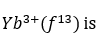Correct option is A
The magnetic moment
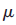
of many d-metal ions can be calculated by using the spin-only approximation because the strong ligand field quenches the orbital contribution.
In a free atom or ion, both the orbital and the spin angular momenta give rise to a magnetic moment and contribute to the paramagnetism. When the atom or ion is part of a complex, any orbital angular momentum is normally quenched, or suppressed, as a result of the interactions of the electrons with their nonspherical environment. However, if any electrons are unpaired the net electron spin angular momentum survives and gives rise to spin-only paramagnetism, which is characteristic of many d-metal complexes.
For orbital angular momentum to contribute, and hence for the paramagnetism to differ significantly from the spin-only value, there must be one or more unfilled or half-filled orbitals similar in energy to the orbitals occupied by the unpaired spins and of the appropriate symmetry (one that is related to the occupied orbital by rotation round the direction of the applied field). If that is so, the applied magnetic field can force the electrons to circulate around the metal ion by using the low-lying orbitals and hence it generates orbital angular momentum and a corresponding orbital contribution to the total magnetic moment.
For the lanthanoids, where the spin-orbital coupling is strong, the orbital angular momentum contributes to the magnetic moment, and the ions behave like almost free atoms. Therefore, the magnetic moment must be expressed in terms of the total angular momentum quantum number J:
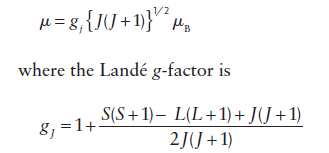
Information Booster
A term symbol is an abbreviated description of the total spin and orbital angular momentum quantum numbers of the electrons in a multi-electron atom.
The usual atomic term symbols assume LS coupling (also known as Russell–Saunders coupling), in which the atom's total spin quantum number S and the total orbital angular momentum quantum number L are "good quantum numbers". The spin-orbit interaction then couples the total spin and orbital moments to give the total electronic angular momentum quantum number J. Atomic states are then well described by term symbols of the form:

where
· S is the total spin quantum number for the atom's electrons. The value 2 S + 1 written in the term symbol is the spin multiplicity, which is the number of possible values of the spin magnetic quantum number MS for a given spin S.
· J is the total angular momentum quantum number for the atom's electrons. J has a value in the range from | L − S| to L + S. The value of J for the ground state is given by (L-S) for a sub-shell that is less than half-filled, and by (L+S) for a sub-shell that is more than half-filled.
L is the total orbital quantum number in spectroscopic notation, in which the symbols for L are: "S", "P", "D", or "F" which represent L = 0, 1, 2, or 3 respectively.