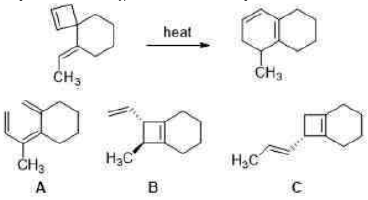
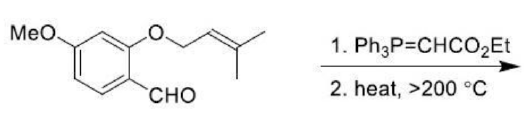
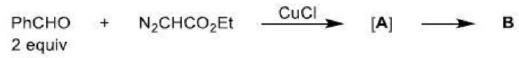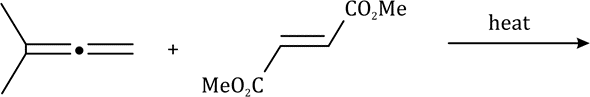Correct option is D
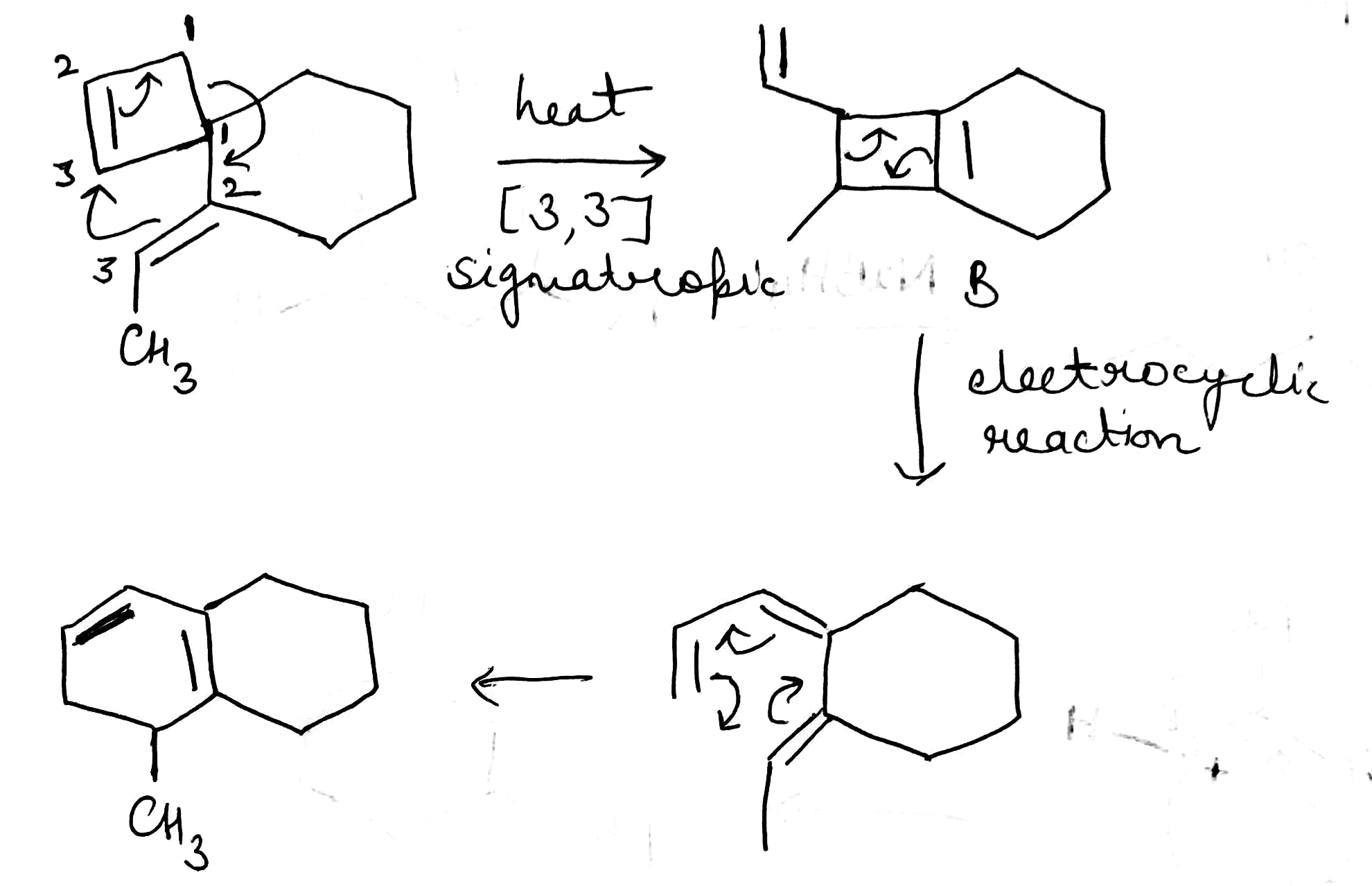
A sigmatropic reaction is a pericyclic reaction wherein the net result is one sigma bond (σ-bond) is changed to another σ-bond in an intramolecular reaction. In this type of rearrangement reaction, a substituent moves from one part of a π-system to another part with simultaneous rearrangement of the π-system.
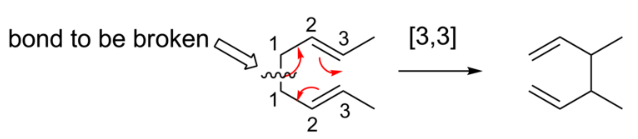
Sigmatropic rearrangements are concisely described by an order term [i,j], which is defined as the migration of a σ-bond adjacent to one or more π systems to a new position (i−1) and (j−1) atoms removed from the original location of the σ-bond.
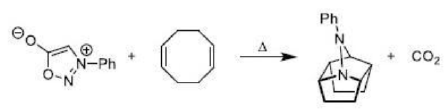
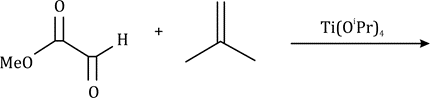
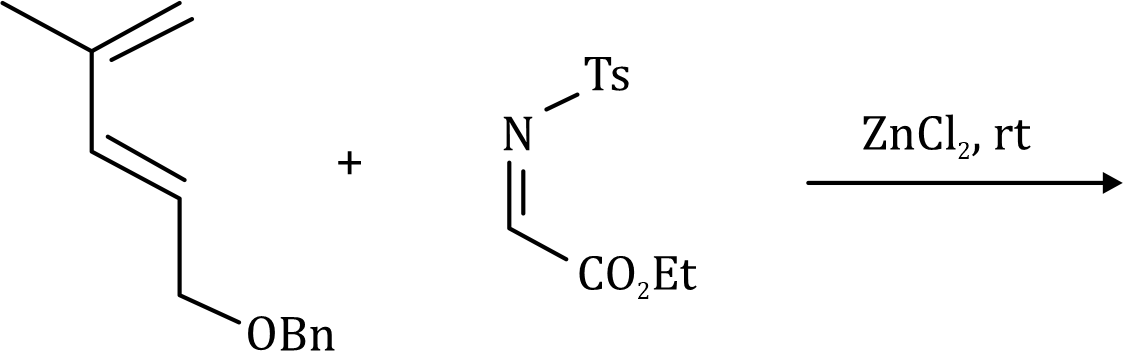
The most important sigmatropic rearrangement are the [3, 3] process involving carbon-carbon bond. The thermal rearrangement of 1, 5-dienes by [3, 3] sigmatropy is called Cope rearrangement. This particular reaction is called a [3, 3] sigmatropic rearrangement because the new σ bond has a 3, 3 relationship to the old σ (sigma) bond.
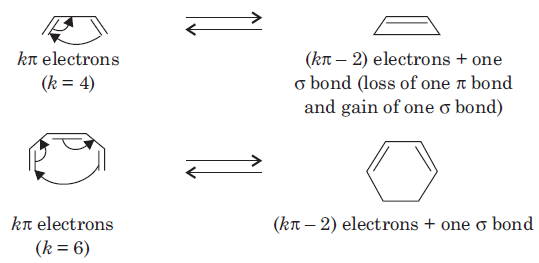
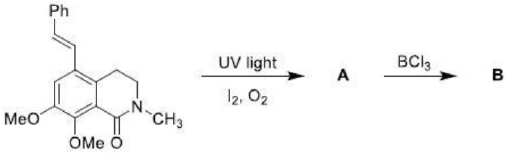
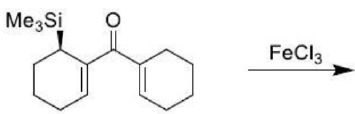
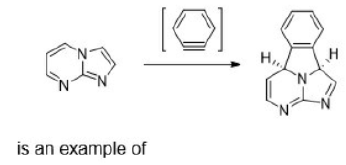
An electrocyclic reaction is the concerted interconversion of a conjugated polyene and a cycloalkene. Electrocyclic reactions are induced either thermally or photochemically. In electrocyclic reactions either a ring is formed with the generation of a new σ bond and the loss of a π bond (i.e., gain of one σ bond and loss of one π bond) or ring is broken with the loss of one σ bond and gain of one π bond.