Correct option is C
Allylic compounds with a good leaving group can form pi-allyl cation complex of palladium (0) via nucleophilic displacement reaction with controlled stereochemistry and regiochemistry. The pi-allyl cation complex gives the product upon attack by a nucleophile. An example is shown below:
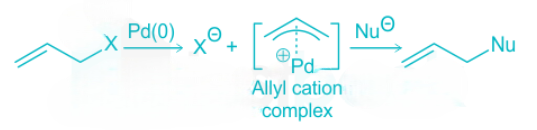
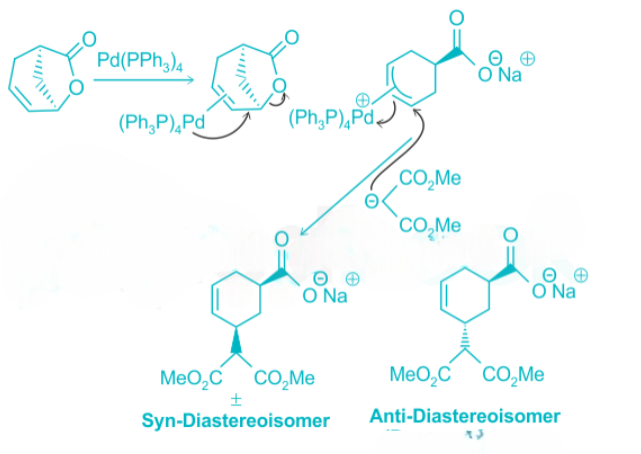
From the above reaction, we can see that the reaction proceeds with the retention of configuration at the reacting center. It also suggests a double inversion.
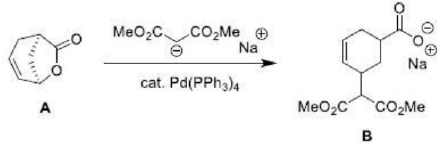
The Coordination of Pd occurs to the double bond of the allylic lactone A on the less hindered face opposite to the leaving group. Also, we may think of the oxidative addition step as an invertive nucleophilic displacement of the leaving group by a pair of Pd electrons. In the last step, the nucleophile then adds to the face of the pi-allyl Pd cation complex opposite of the Pd metal.
This results in the formation of a syn-diastereomer B, which is racemic.