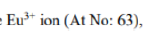Correct option is D
In general, the experimental values of bulk magnetic moments of Ln3+ ions agree well with the calculated ones. This is based on the assumption of Russell–Saunders coupling and large spin–orbit coupling constants, as a consequence of which only the states of lowest J value are
populated. This is not true for Eu3+. For Eu3+(f6) the spin–orbit coupling constant λ is
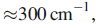
only slightly greater than
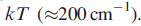
The ground state of the f6 ion is 7F0 (which is diamagnetic, since J=0), but the states
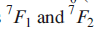
are also populated to some extent and give rise to the observed magnetic moment. As expected, at low temperatures, the moment of Eu3+approaches zero.
The 4f orbital in Eu³⁺ is not more than half-filled. The electron configuration for Eu³⁺ is 4f⁶, which is less than half-filled. A half-filled 4f orbital would require 7 electrons, which is not the case here.