Correct option is B
The notation for a full term symbol is:
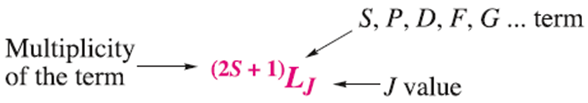
The energy and the orbital angular momentum of a multielectron species are determined by a quantum number, L. Energy states for which L=0, 1, 2, 3, 4... are known as S, P, D, F, G... terms, respectively.
For any system containing more than one electron, the energy of an electron with principal quantum number n depends on the value of l, and this also determines the orbital angular momentum which is given by the equation:

The spin quantum number, s, determines the magnitude of the spin angular momentum of an electron and has a value of 1/2. For a 1-electron species, ms is the magnetic spin angular momentum and has a value of +1/2 or -1/2.The spin angular momentum for a multielectron species is given by the following equation, where S is the total spin quantum number.
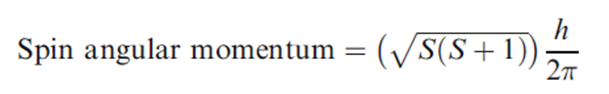
The quantum number MS is obtained by algebraic summation of the ms values for individual electrons:
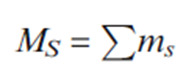
For each value of S, there are (2S+1) values of MS.

The interaction between the total angular orbital momentum, L, and the total spin angular momentum, S is defined by the total angular momentum quantum number, J.
The following equation gives the relationship for the total angular momentum for a multi-electron species.

The value of J for the ground state is given by (L-S) for a sub-shell that is less than half-filled, and by (L+S) for a sub-shell that is more than half-filled.
For the d3 electronic configuration. The d orbital is less than half filled.
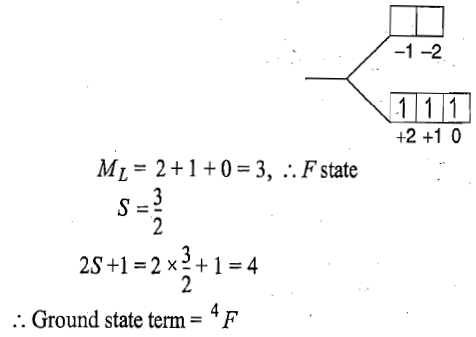
J=3-3/2=3/2







