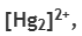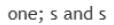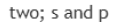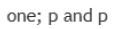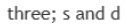Correct option is A
Mercury is a chemical element; it has symbol Hg and atomic number 80.Mercury is a heavy, silvery-white metal that is liquid at room temperature. Compared to other metals, it is a poor conductor of heat, but a fair conductor of electricity.
Most mercury(I) compounds are diamagnetic and feature the dimeric cation, Hg22+. Stable derivatives include the chloride and nitrate. In aqueous solution of a mercury(I) salt, slight disproportion of Hg22+ into Hg and Hg2+ results in >0.5% of dissolved mercury existing as Hg2+.In these solutions, complexation of the Hg2+with addition of ligands such as cyanide causes disproportionation to go to completion, with all Hg22+precipitating as elemental mercury and insoluble mercury(II) compounds (e.g. mercury(II) cyanide if cyanide is used as the ligand). Mercury(I) chloride, a colorless solid also known as calomel, is really the compound with the formula Hg2Cl2, with the connectivity Cl-Hg-Hg-Cl. It reacts with chlorine to give mercury(II) chloride, which resists further oxidation. Mercury(I) hydride, a colorless gas, has the formula HgH, containing no Hg-Hg bond; however, the gas has only ever been observed as isolated molecules.
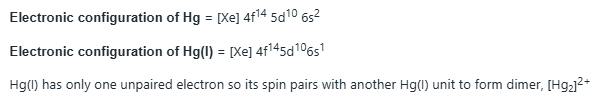
s orbitals are involved in bonding.