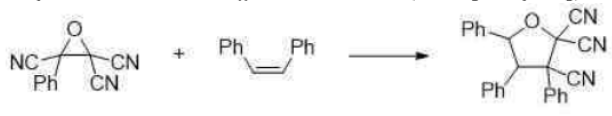
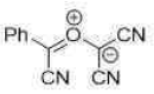
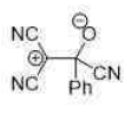
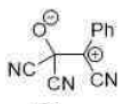
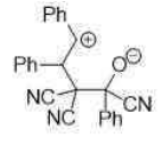
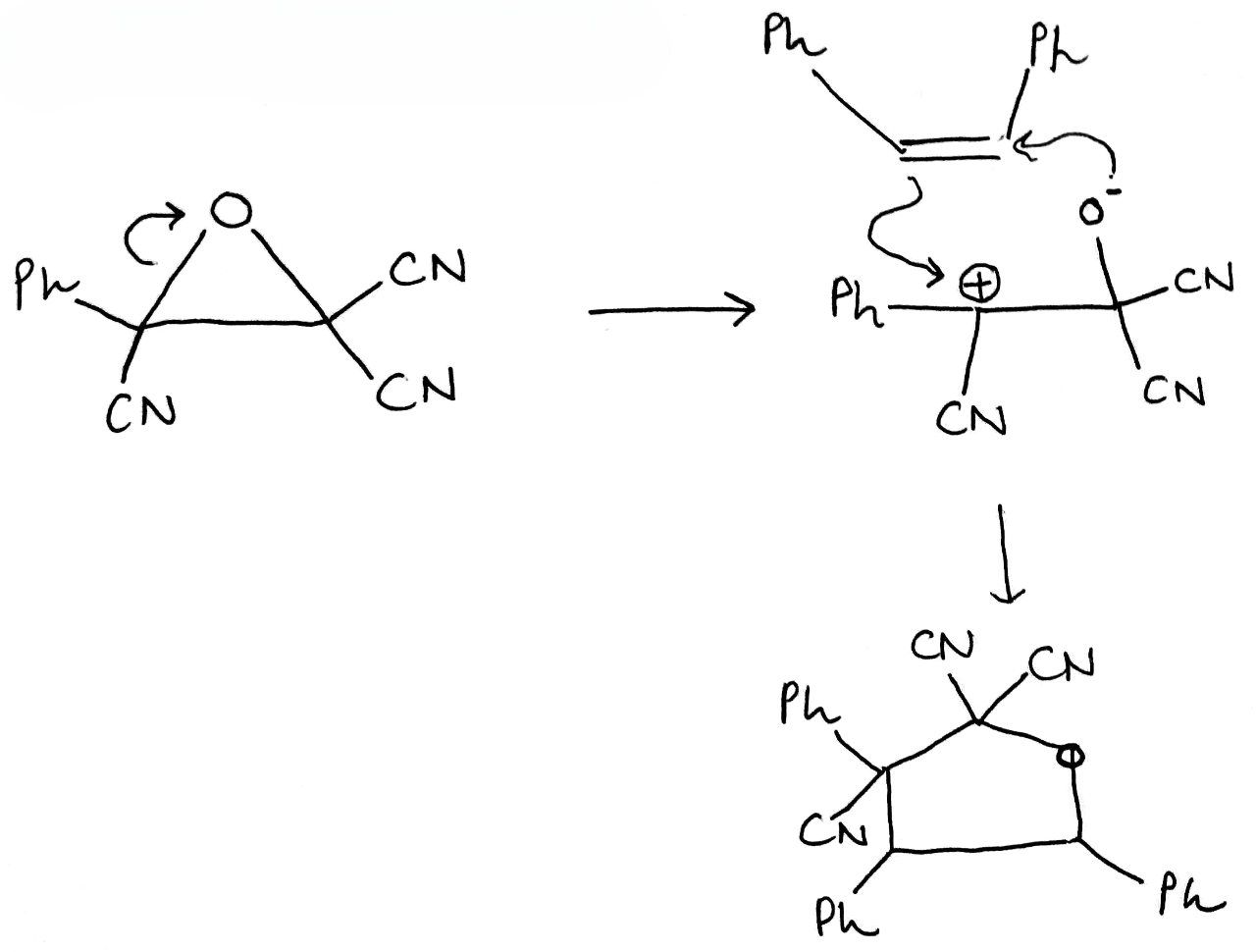
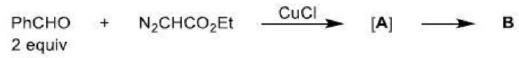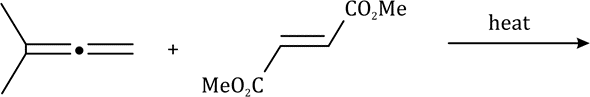Correct option is C
An electron-withdrawing group (EWG; here CN) is a group or atom that has the ability to draw electron density toward itself and away from other adjacent atoms. This electron density transfer is often achieved by resonance or inductive effects.
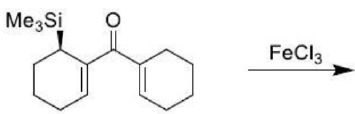
In the given reaction, on opening the three membered ring, positive charge will be stabilized by the Ph group (due to delocalization of the positive charge in the ring).
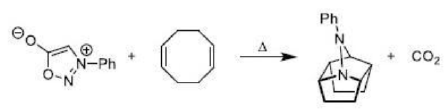
There is a large class of reactions, known as 1, 3-dipolar cycloaddition reactions. These reactions can be represented as in the given equation:
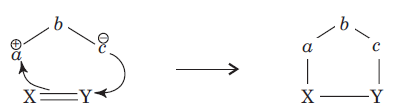
The species
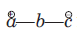
is called the 1, 3-dipolar molecule and X=Y is the dipolarophile. The 1, 3-dipolar molecules are isoelectronic with the allyl carbanion and have four electrons in a π (pi) system encompassing the 1, 3-dipole. All 1, 3-dipoles contain 4π (pi) electrons in three parallel p orbitals of a, b and c.
In 1, 3-dipolar species
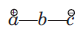
i) a has six electrons in its outermost orbit. b has its complete octet having at least one lone pair of electrons. c has its complete octet having negative charge.
ii)a may be carbon, oxygen or nitrogen, b may be nitrogen or oxygen, and c may be carbon, oxygen or nitrogen.
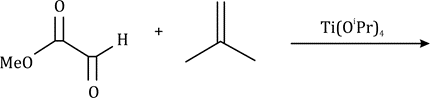
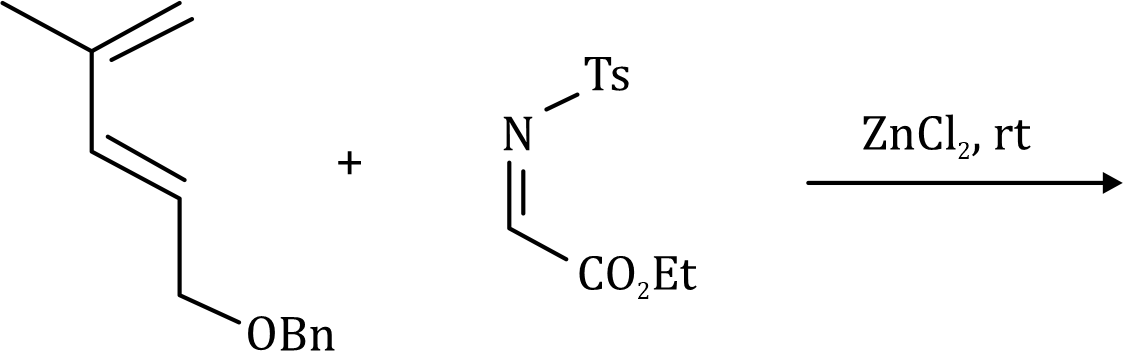
The dipolarophiles (X=Y) are alkenes, alkynes, imines, nitriles and carbonyl compounds.