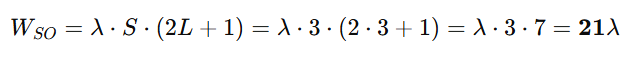Correct option is B
To calculate the multiplet width (WSO) for Eu³⁺, we use the formula:
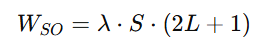
Europium (Z = 63) in its neutral state has configuration [Xe]4f⁷6s². Upon losing 3 electrons to form Eu³⁺, its configuration becomes: [Xe] 4f⁶
Number of unpaired electrons = 6 (for 4f⁶)
Maximum multiplicity implies S = 3 (since each unpaired electron contributes ½ spin)
The total orbital angular momentum (L) for 4f⁶ is L = 3 (based on standard term symbol for f⁶ configuration)
Now apply the formula: