Correct option is B
CO molecule
The carbon atom has 2+4=6 electrons, and the O atom has 2+6=8 electrons, so the CO molecule contains 14 electrons.
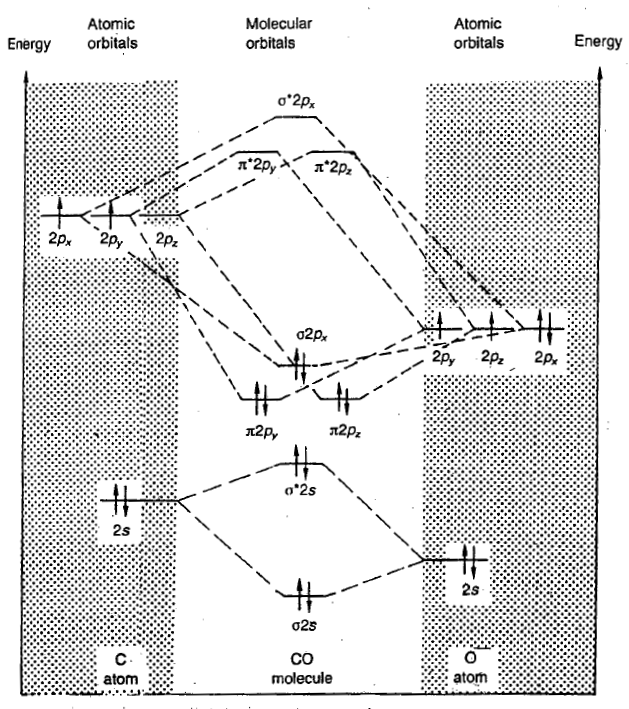
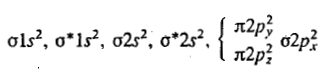
In chemistry, pi backbonding or π backbonding is a π-bonding interaction between a filled (or half filled) orbital of a transition metal atom and a vacant orbital on an adjacent ion or molecule. The ligands involved in π backbonding can be broken into three groups: carbonyls and nitrogen analogs, alkenes and alkynes, and phosphines. The electrons are partially transferred from a d-orbital of the metal to anti-bonding molecular orbitals of CO (and its analogs). This electron-transfer strengthens the metal–C bond and weakens the C–O bond. The strengthening of the M–CO bond is reflected in increases of the vibrational frequencies for the M–C bond. Furthermore, the M–CO bond length is shortened. The weakening of the C–O bond is indicated by a decrease in the wavenumber of the νCO band(s) from that for free CO (2143 cm-1).
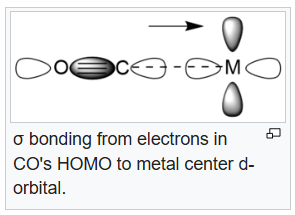
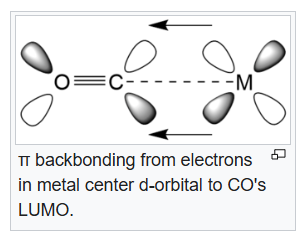
In the Lewis acid-base adduct (CF3)3B.CO, (CF3)3B acts as a Lewis acid and CO acts as a base. Donation of electrons from CO's HOMO will result in an increase in bond order and bond strength which results in a higher CO stretching frequency. Thus, the CO stretching frequency in IR will be more than 2143 cm-1 (in free CO). The 19F NMR spectrum of (CF3)3B·CO does not show a singlet resonance.
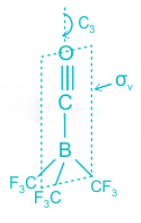
The point group of (CF3)3B.CO is C3v. The molecule (CF3)3B.CO, contains a C3 proper axis of symmetry and one σv plane.