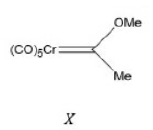
Correct option is C
Group 6, numbered by IUPAC style, is a group of elements in the periodic table. Its members are chromium (Cr), molybdenum (Mo), and tungsten (W).
Early d-block metal complexes containing one or two alpha-hydrogen atoms may undergo alpha-hydrogen abstraction to yield carbene (alkylidene) or carbyne (alkylidyne) complexes.

Carbenes can be made by nucleophilic attack on a carbonyl C atom followed by alkylation.
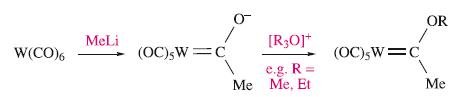
Compounds of the type formed in the above reaction are called Fischer-type carbenes. They possess a low oxidation state metal, a heteroatom (O in this example) and an electrophilic carbene centre (i.e. subject to attack by nucleophiles).

Resonance pair gives a bonding description for a Fischer-type carbene complex.
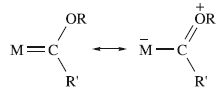
The rotational barrier across the metal and carbene carbon is low as there is no complete double bond present.
A Fischer carbene is a type of transition metal carbene complex, which is an organometallic compound containing a divalent organic ligand. In a Fischer carbene, the carbene ligand is a σ-donor π-acceptor ligand. Because π-backdonation from the metal centre is generally weak, the carbene carbon is electrophilic.
Structure
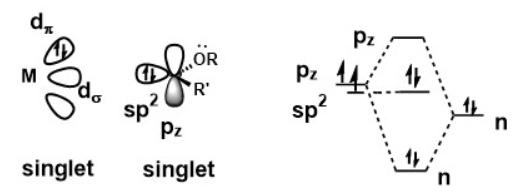
A metal carbene complex could be considered a Fischer carbene when the carbene is in singlet state. Delocalization of the lone pair from the substituent on carbene carbon raises the energy of pz orbital, thus forcing the two of electrons of carbene stay as an electron pair. Bonding between carbene and the metal centre involves a strong σ donation from sp2 orbital to an empty d orbital on metal centre and a weak π back donation from the metal centre to the empty pz orbital. Because the π donation is weak, the carbene carbon is electrophilic in nature.