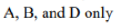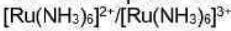Correct option is D
Bromine trifluoride is a pale yellow liquid at 298K and is an aprotic non-aqueous solvent. Bromine trifluoride is an extremely powerful fluorinating agent and fluorinates essentially every species that dissolves in it. However, massive quartz is kinetically stable towards BrF3 and the solvent can be handled in quartz vessels. Apparatus made from Cu, Ni or Monel metal (68% Ni and 32% Cu) can also be used; the metal surface becomes protected by a thin layer of metal fluoride.
The self-ionization of BrF3 has been demonstrated by the isolation and characterization of salts containing the [BrF2]+and [BrF4]- ions, and by conductometric titrations of them. Using the solvent-based acid– base definitions, an acid in BrF3 is a species that produces [BrF2]+, and a base is one that gives [BrF4]-.
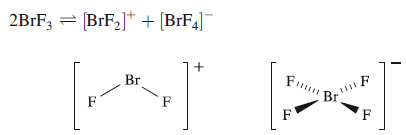
X,Y and Z have two lone pairs of electrons.
Y has a bent shape
Z has a square planar shape