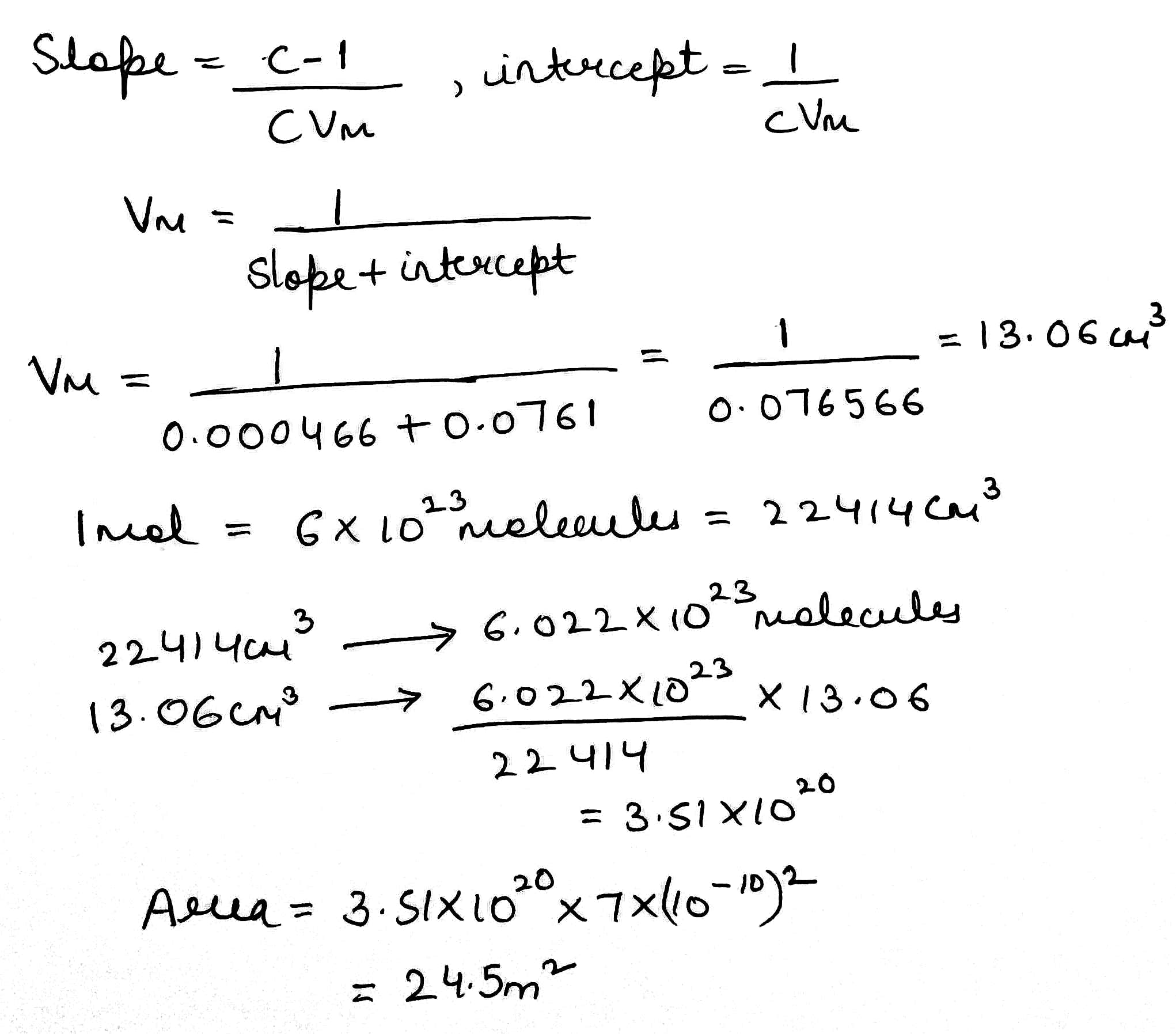
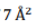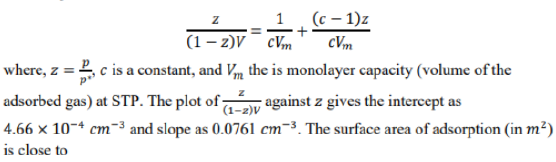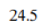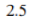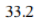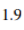Correct option is A
Multilayer Adsorption
The adsorption of gases on the surface of an adsorbent is no more monolayer at high pressures and low temperatures. At high pressure, the number of molecules striking per unit area of the surface per unit time is quite high. On the other hand, at low temperature the thermal energy of molecules is not sufficiently large to overcome the forces of attraction (van der Waals forces of attraction) between the adsorbed molecule and nearby unadsorbed molecule. This results into the multilayer adsorption, i.e. more than one layer of molecules is adsorbed at the surface. The formation of multilayer is very much enhanced as the pressure of the gas reaches near to the saturation vapour pressure of the liquefied adsorbent at the given experimental temperature.
The adsorption isotherms shown in figure have been interpreted by Brunauer, Emmett and Teller on the basis of formation of multilayer. They derived a theoretical expression, known as BET adsorption isotherm. While deriving the expression, it was assumed that interactions amongst the adsorbed molecules in the adsorption layer along the adsorbent surface are neglected.