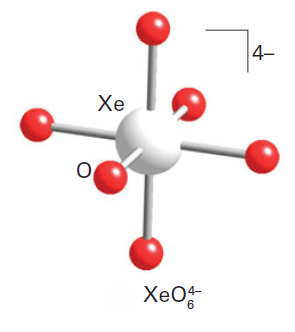
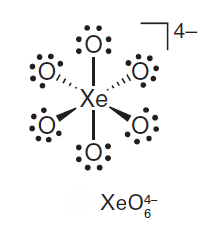
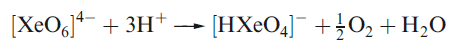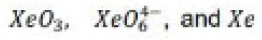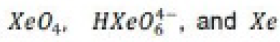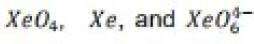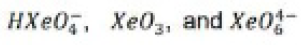Correct option is A
The most stable Xe compounds are the colourless fluorides XeF2, XeF4 and XeF6. XeF6 always forms with contamination by the lower fluorides. Separation of XeF4 from a mixture involves preferential complexation of XeF2 and XeF6 and the XeF4 is then removed in vacuo, while separation of XeF6 involves reaction followed by thermal decomposition of the complex.
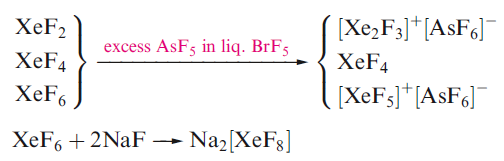
All the fluorides sublime in vacuo, and all are readily decomposed by water.
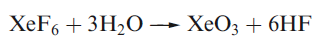
Solid XeO3 forms colourless crystals and is dangerously explosive
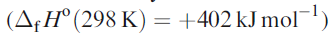
The solid contains trigonal pyramidal molecule. Xenon trioxide is only weakly acidic and its aqueous solution is virtually non-conducting. Reactions of XeO3 and MOH (M= K, Rb, Cs) produce xenates which slowly disproportionate in solution.
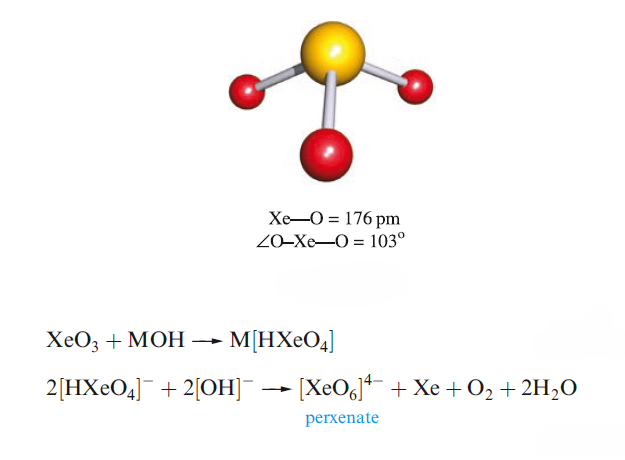
Aqueous [XeO6]4- is formed when O3 is passed through a dilute solution of XeO3 in alkali. Insoluble salts such as Na4XeO6.8H2O and Ba2XeO6 may be precipitated, but perxenic acid ‘H4XeO6’ (a weak acid in aqueous solution) has not been isolated. The perxenate ion is a powerful oxidant and is rapidly reduced in aqueous acid. Oxidations such as Mn (II) to [MnO4]- occur instantly in acidic media at 298 K.