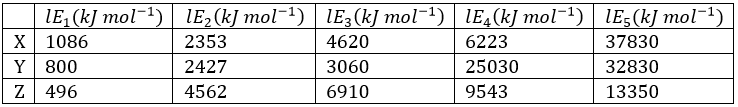
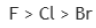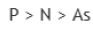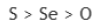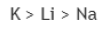Correct option is C
The electron affinity (Eea) of an atom or molecule is defined as the amount of energy released when an electron attaches to a neutral atom or molecule in the gaseous state to form an anion.
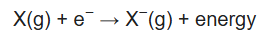
Atoms whose anions are more stable than neutral atoms have a greater Eea. Chlorine most strongly attracts extra electrons; neon most weakly attracts an extra electron.
Eea generally increases across a period (row) in the periodic table prior to reaching group 18. This is caused by the filling of the valence shell of the atom; a group 17 atom releases more energy than a group 1 atom on gaining an electron because it obtains a filled valence shell and therefore is more stable. In group 18, the valence shell is full, meaning that added electrons are unstable, tending to be ejected very quickly.
Counterintuitively, Eea does not decrease when progressing down most columns of the periodic table.
Option a belongs to group 17
Option b belongs to group 15
Option c belongs to group 16
Option d belongs to group 1