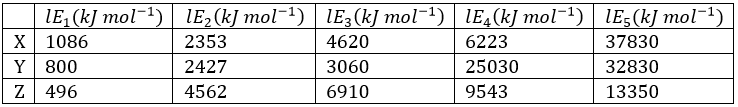
Correct option is C
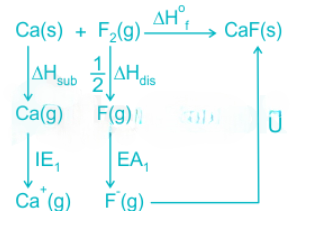
The Born–Haber cycle is an approach to analyze reaction energies. The cycle is concerned with the formation of an ionic compound from the reaction of a metal (often a Group I or Group II element) with a halogen or other non-metallic element such as oxygen.
Born–Haber cycles are used primarily as a means of calculating lattice energy, which cannot otherwise be measured directly. The lattice enthalpy is the enthalpy change involved in the formation of an ionic compound from gaseous ions (an exothermic process), or sometimes defined as the energy to break the ionic compound into gaseous ions (an endothermic process). A Born–Haber cycle applies Hess's law to calculate the lattice enthalpy by comparing the standard enthalpy change of formation of the ionic compound (from the elements) to the enthalpy required to make gaseous ions from the elements.
This lattice calculation is complex. To make gaseous ions from elements it is necessary to atomise the elements (turn each into gaseous atoms) and then to ionise the atoms. If the element is normally a molecule then we first have to consider its bond dissociation enthalpy. The energy required to remove one or more electrons to make a cation is a sum of successive ionization energies; for example, the energy needed to form Mg2+ is the ionization energy required to remove the first electron from Mg, plus the ionization energy required to remove the second electron from Mg+. Electron affinity is defined as the amount of energy released when an electron is added to a neutral atom or molecule in the gaseous state to form a negative ion.
In the hypothetical compound NaCl2, the constituent ions are Na2+and 2Cl-.
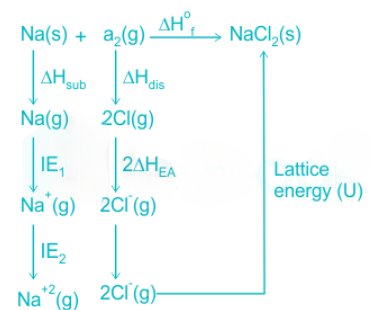
The very high positive value of IE2 for the Na atom is not compensated by the high lattice energy value of NaCl2.
In the hypothetical compound CaF, the constituent ions are Ca+and F-.