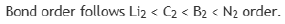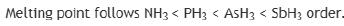Correct option is D
The bond order of a molecule is a concept in chemistry that indicates the strength or stability of a chemical bond between two atoms within a molecule.
C2 comprises of twelve electrons.
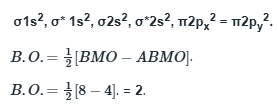
B2 comprises of ten electrons
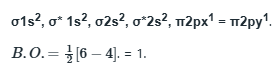
N2 comprises of 14 electrons

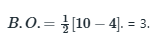
Li2 comprises of six electrons
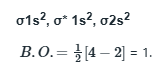
More is the number of electrons in the anti-bonding molecular orbital, lesser is the stability.

The melting point of a substance is the temperature at which it changes state from solid to liquid. At the melting point the solid and liquid phase exist in equilibrium.
Melting point increases down the group. On going down the group ,atomic size increases which leads to increase in the Vander Waal's forces. NH3 shows high melting point due to the presence of intermolecular hydrogen bonding. Hence, the order of melting point is PH3<AsH3<SbH3<NH3
Electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Electronegativity increases on passing from left to right along a period. On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more "pull" it will have on electrons) and the number and location of other electrons in the atomic shells (the more electrons an atom has, the farther from the nucleus the valence electrons will be, and as a result, the less positive charge they will experience—both because of their increased distance from the nucleus and because the other electrons in the lower energy core orbitals will act to shield the valence electrons from the positively charged nucleus).

Ionization energy (IE) is the minimum energy required to remove the most loosely bound electron of an isolated gaseous atom, positive ion, or molecule. The first ionization energy is quantitatively expressed as
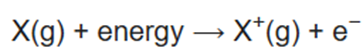
where X is any atom or molecule, X+ is the resultant ion when the original atom was stripped of a single electron, and e− is the removed electron. Roughly speaking, the closer the outermost electrons are to the nucleus of the atom, the higher the atom's ionization energy. Ionization energy generally increases from left to right within a given period (that is, row). As the nuclear charge of the nucleus increases across the period, the electrostatic attraction increases between electrons and protons, hence the atomic radius decreases, and the electron cloud comes closer to the nucleus because the electrons, especially the outermost one, are held more tightly by the higher effective nuclear charge.

It is easier to remove an electron from p orbital than to remove an electron from fully filled s orbital.