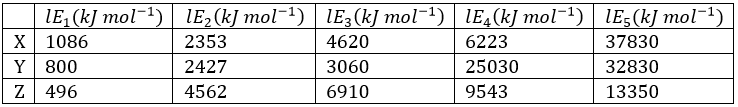
Correct option is D
The compounds which are soluble in water and the one which is insoluble in water depends upon the solubility of that particular substance.
Sulphides of As(III) and Sb(III) form polysulphide complex hence soluble in yellow ammonium sulphide.
The solubility product of PbCl2 is 1.75 × 10-5 at a normal temperature.
The solubility product shows that lead chloride is less soluble in water.
As(III) and Sb (III) are not soluble in normal sulphide
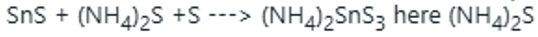
is yellow ammonium sulphide. So, it is soluble in yellow ammonium sulphide.
When MnS is treated with HgS the solubility of Mn2+ is more than Hg2+ hence, it does not form any precipitate.