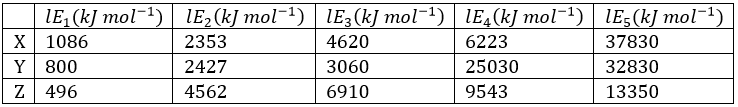
Correct option is B
Diborane, commonly known as diborane, is the chemical compound with the formula B2H6. It is a highly toxic, colorless, and pyrophoric gas with a repulsively sweet odor. Given its simple formula, borane is a fundamental boron compound. It has attracted wide attention for its electronic structure. Several of its derivatives are useful reagents.
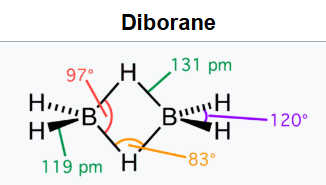
The structure of diborane has D2h symmetry. Four hydrides are terminal, while two bridge between the boron centers. The lengths of the B–H bridge bonds and the B–H terminal bonds are 1.33 and 1.19 Å respectively. This difference in bond lengths reflects the difference in their strengths, the B–H bridge bonds being relatively weaker. The weakness of the B–H bridge compared to B–H terminal bonds is indicated by their vibrational signatures in the infrared spectrum, being ≈2100 and 2500 cm-1 respectively.
The model determined by molecular orbital theory describes the bonds between boron and the terminal hydrogen atoms as conventional 2-center 2-electron covalent bonds. The bonding between the boron atoms and the bridging hydrogen atoms is, however, different from that in molecules such as hydrocarbons. Each boron uses two electrons in bonding to the terminal hydrogen atoms and has one valence electron remaining for additional bonding. The bridging hydrogen atoms provide one electron each. The B2H2 ring is held together by four electrons forming two 3-center 2-electron bonds. This type of bond is sometimes called a "banana bond".
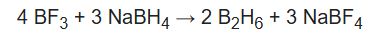
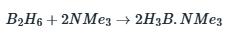
Boranes are diamagnetic compounds that are colorless in nature and have low thermal stability. Diborane is a sweet-smelling gas that is highly poisonous at room temperature. It is also highly reactive. It generates higher boranes at higher temperatures, releasing hydrogen.