Correct option is C
In a high spin octahedral complex of d6metal ion, the d orbitals split into t2g (lower energy) and eg orbitals (higher energy) as shown in figure
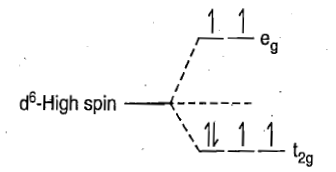
Transitions of unpaired electron from t2g orbitals to eg orbitals are restricted because there will be change in spin multiplicity that is these transitions are spin forbidden. Transition of that electron in t2g orbital which is paired and has the opposite spin to all other electrons can occur because this transition is spin allowed. Since there is transition of only one electron, therefore, this transition is similar to transition in d1 octahedral complexes.
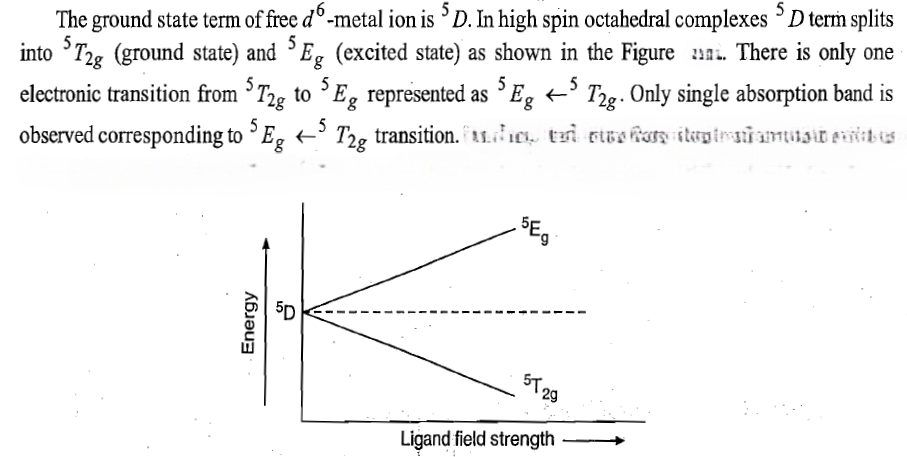
The ground state term of d6 metal ion can be found as:
S=4(1/2)=2.
Spin multiplicity: 2S+1=2(2)+1=5
L=2 [+2+2+1+0-1-2]
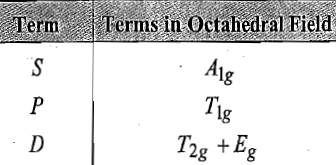
The spin only formula for magnetic moment is given as

For d6 metal ion, n=4, thus magnetic moment is 4.89 B.M.







