Correct option is B
The oxide UO3 is polymorphic and all forms decompose to the mixed oxidation state U3O8 on heating. Most acids dissolve UO3 to give yellow solutions containing the uranyl ion, present as a complex, e.g. in aqueous solution, exists as an aqua ion, and the perchlorate
salt of the pentagonal bipyramidal
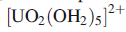
has been isolated.
The [UO2]2+ ion is present in many solid compounds including the alkaline earth uranates (e.g. BaUO4) which are best described as mixed metal oxides. In aqueous solution, the [UO2]2+ ion is partially hydrolysed to species such as [U2O5]2+and [U3O8]2+. In aqueous alkaline solution, the species present depend on the concentrations of both [UO2]2+and OH-.The [UO2]2+ ion is hard and forms a more stable complex with F- than with the later halides.
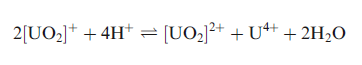
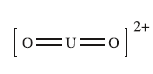
Both the 5f and 6d orbitals of U are involved in bonding with the 2p orbitals of oxygen atom.
The uranyl ion is linear and symmetrical, with both U–O bond lengths of about 180 pm. The bond lengths are indicative of the presence of multiple bonding between the uranium and oxygen atoms. Since uranium(VI) has the electronic configuration of the preceding noble gas, radon, the electrons used in forming the U–O bonds are supplied by the oxygen atoms. The electrons are donated into empty atomic orbitals on the uranium atom. The empty orbitals of lowest energy are 7s, 5f and 6d.






