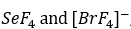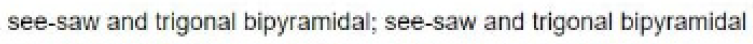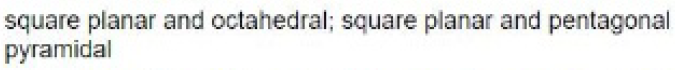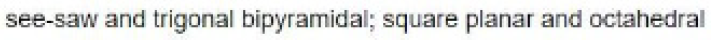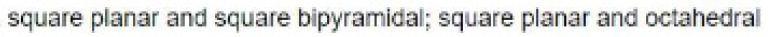Correct option is C
Valence shell electron pair repulsion (VSEPR) theory is a model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms. The premise of VSEPR is that the valence electron pairs surrounding an atom tend to repel each other. The greater the repulsion, the higher in energy (less stable) the molecule is. Therefore, the VSEPR-predicted molecular geometry of a molecule is the one that has as little of this repulsion as possible.
VSEPR theory is used to predict the arrangement of electron pairs around central atoms in molecules, especially simple and symmetric molecules. A central atom is defined in this theory as an atom which is bonded to two or more other atoms, while a terminal atom is bonded to only one other atom.
The overall geometry is further refined by distinguishing between bonding and nonbonding electron pairs. The bonding electron pair shared in a sigma bond with an adjacent atom lies further from the central atom than a nonbonding (lone) pair of that atom, which is held close to its positively charged nucleus. VSEPR theory therefore views repulsion by the lone pair to be greater than the repulsion by a bonding pair. As such, when a molecule has 2 interactions with different degrees of repulsion, VSEPR theory predicts the structure where lone pairs occupy positions that allow them to experience less repulsion. Lone pair–lone pair (lp–lp) repulsions are considered stronger than lone pair–bonding pair (lp–bp) repulsions, which in turn are considered stronger than bonding pair–bonding pair (bp–bp) repulsions, distinctions that then guide decisions about overall geometry when 2 or more non-equivalent positions are possible
The electronic geometry of a molecule depends on the number of bond pairs and lone pairs, while the shape depends only on the number of bond pairs.
The steric number of a central atom in a molecule is the number of atoms bonded to that central atom, called its coordination number, plus the number of lone pairs of valence electrons on the central atom.
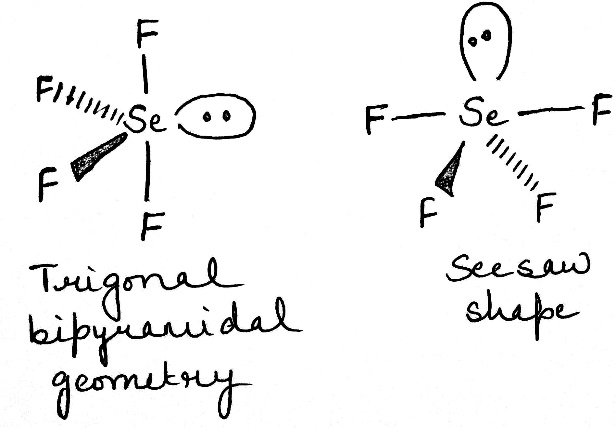
SeF4
In the molecule SeF4 , for example, the central selenium atom has four ligands; the coordination number of selenium is four. In addition to the four ligands, selenium also has one lone pair in this molecule. Thus, the steric number is 4 + 1 = 5. When 5 valence electron pairs surround a central atom, they adopt a trigonal bipyramidal molecular geometry with two collinear axial positions and three equatorial positions. An electron pair in an axial position has three close equatorial neighbors only 90° away and a fourth much farther at 180°, while an equatorial electron pair has only two adjacent pairs at 90° and two at 120°. The repulsion from the close neighbors at 90° is more important, so that the axial positions experience more repulsion than the equatorial positions; hence, when there are lone pairs, they tend to occupy equatorial positions as shown in the diagrams of the next section for steric number five.
The seesaw molecular shape is found in selenium tetrafluoride ( SeF4). The name "seesaw" comes from the observation that it looks like a playground seesaw. The seesaw shape occurs when a molecule has a steric number of 5, with the central atom being bonded to 4 other atoms and 1 lone pair. An equatorial lone pair is repelled by only two bonding pairs at 90°, whereas a hypothetical axial lone pair would be repelled by three bonding pairs at 90° which would make the molecule unstable. Repulsion by bonding pairs at 120° is much smaller and less important.
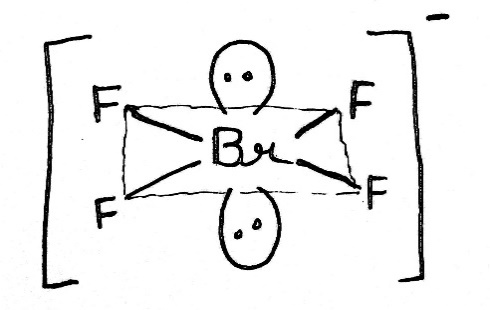
[BrF4]-
In the molecule [BrF4]- , for example, the central bromine atom has four ligands; the coordination number of bromine is four. In addition to the four ligands, bromine also has two lone pairs in this molecule. Thus, the steric number is 4 + 2=6. When 6 valence electron pairs surround a central atom, they adopt an octahedral molecular geometry. Octahedral molecular geometry also called square bipyramidal describes the shape of compounds with six atoms or groups of atoms or ligands symmetrically arranged around a central atom, defining the vertices of an octahedron. The bond angle is 90 degrees.
The square planar shape occurs when a molecule has a steric number of 6, with the central atom being bonded to 4 other atoms and 2 lone pairs.
Additional Knowledge
In geometry, a pentagonal pyramid is a pyramid with a pentagon base and five triangular faces, having a total of six faces.