Correct option is B
In the given case, the extraction efficiency is determined by the distribution of the solute S between the two phases (aqueous and organic) during the liquid-liquid extraction process. The partition coefficient (KD) is a crucial factor that governs this distribution. KD represents the ratio of the concentration of the solute in one phase to its concentration in the other phase at equilibrium.
In this case, the solute S has a partition coefficient (KD) of five between water and chloroform. This means that at equilibrium, for every one unit of the solute in the aqueous phase, there will be five units of the solute in the chloroform phase.
Mathematically,

Given, a 0.05M aqueous solution of the solute in a 50mL sample, the initial amount of solute in the aqueous phase:
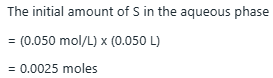
Now, using 15mL of chloroform to extract the solute from the aqueous phase. Before extraction, there is no solute in the chloroform phase. However, during the extraction process, some of the solute will move from the aqueous phase into the chloroform phase based on the KD value.







