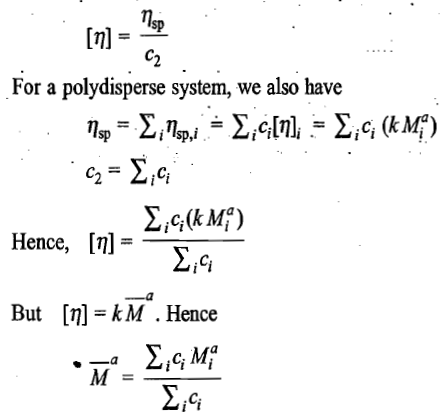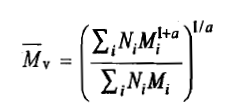Correct option is C
Viscosity is a measure of a fluid's rate-dependent resistance to a change in shape or to movement of its neighboring portions relative to one another. For liquids, it corresponds to the informal concept of thickness; for example, syrup has a higher viscosity than water.
Viscosity quantifies the internal frictional force between adjacent layers of fluid that are in relative motion. For instance, when a viscous fluid is forced through a tube, it flows more quickly near the tube's center line than near its walls. Experiments show that some stress (such as a pressure difference between the two ends of the tube) is needed to sustain the flow. This is because a force is required to overcome the friction between the layers of the fluid which are in relative motion. For a tube with a constant rate of flow, the strength of the compensating force is proportional to the fluid's viscosity.
Viscosity of a polymer solution
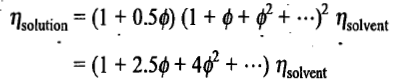
The above equation is known as Einstein's equation for the viscosity of a solution.
While dealing with a polymer solution, the solute concentration is usually expressed in terms of mass per unit volume. We can express volume fraction of solute in terms of mass per unit volume of solution as follows

V is the volume of solution

Definitions of a few terms
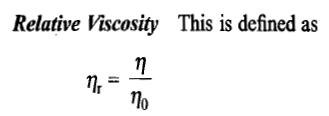
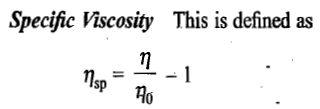
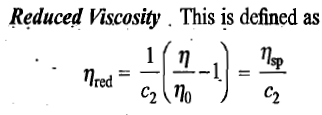
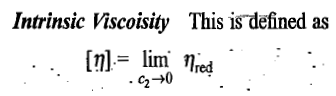
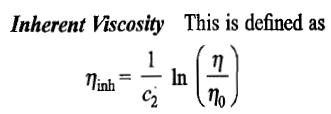
The Mark-Houwink equation
The intrinsic viscosity of a polymer solution is found to increase with increase in the average molar mass of the polymer. This dependence is expressed as
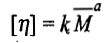
The above equation is known as the Mark-Houwink equation. The constants k and a are known as the Mark-Houwink constants for a system. The values of k and a depend on the (i) nature of polymer, ii) nature of the solvent, and (iii) temperature. Both [η] and k are expressed in the same unit. The molar mass
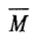
carries the unit of mass used in the concentration term appeared in the quantity [η].
The extensive tabulations of k and a are available in the literature. This follows from the following analysis. Since [η] is a limiting value as c2 → 0, the concentration effect it contains can be written as