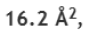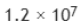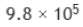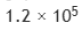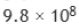Correct option is B
Determination of surface area of adsorbent
Since the molar volume of an ideal gas at STP is
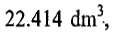
the number of molecules N adsorbed corresponding to the volume
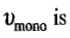
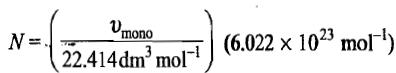
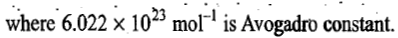
Now if the area of cross-section of a single molecule is known, it can be multiplied by the above number to give the total surface area of the adsorbent.

Assuming the molecule to be spherical, its cross-sectional area can be computed as follows. If r is the radius of the molecule, it follows that
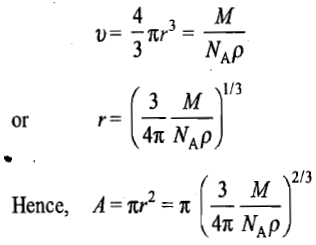
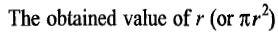
is only an approximate one since it lacks the information regarding the exact nature of packing at the surface of the adsorbent.