Correct option is B
Allyl ligands can donate sigma electrons and pi electrons. They are also good pi acceptors due to the availability of anti-bonding π* orbitals.
The Hückel method or Hückel molecular orbital theory, is a simple method for calculating molecular orbitals as linear combinations of atomic orbitals.
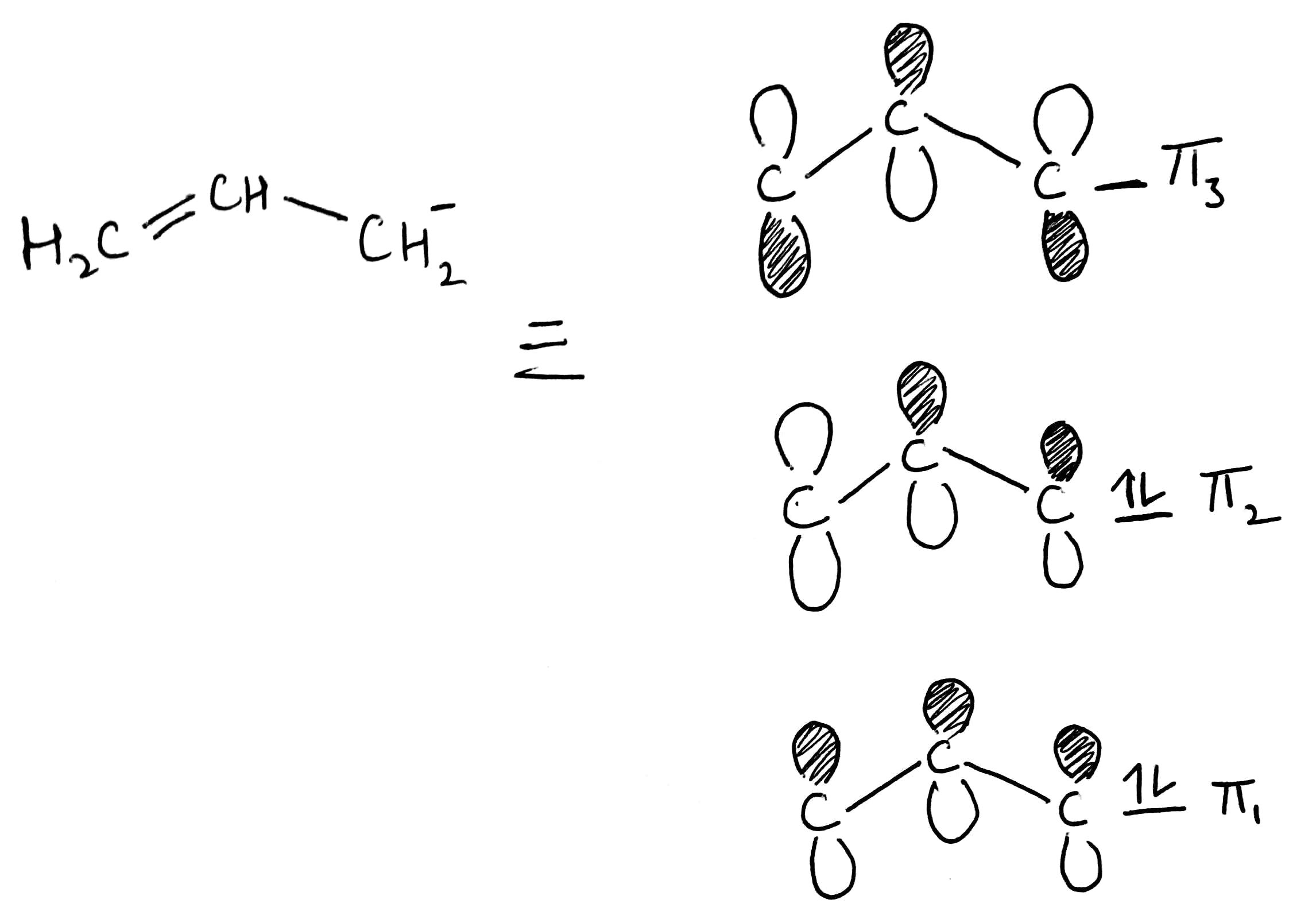

The last orbital which is unoccupied can accept pi electrons.







