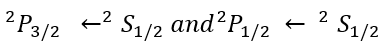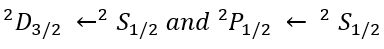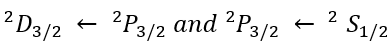Correct option is A
A term symbol is an abbreviated description of the total spin and orbital angular momentum quantum numbers of the electrons in a multi-electron atom.
The usual atomic term symbols assume LS coupling (also known as Russell–Saunders coupling), in which the atom's total spin quantum number S and the total orbital angular momentum quantum number L are "good quantum numbers". The spin-orbit interaction then couples the total spin and orbital moments to give the total electronic angular momentum quantum number J. Atomic states are then well described by term symbols of the form:
2S+1LJ
where
· S is the total spin quantum number for the atom's electrons. The value 2 S + 1 written in the term symbol is the spin multiplicity, which is the number of possible values of the spin magnetic quantum number MS for a given spin S.
· J is the total angular momentum quantum number for the atom's electrons. J has a value in the range from | L − S| to L + S.
L is the total orbital quantum number in spectroscopic notation, in which the symbols for L are: "S", "P", "D", or "F" which represent L = 0, 1, 2, or 3 respectively.