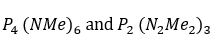Correct option is A
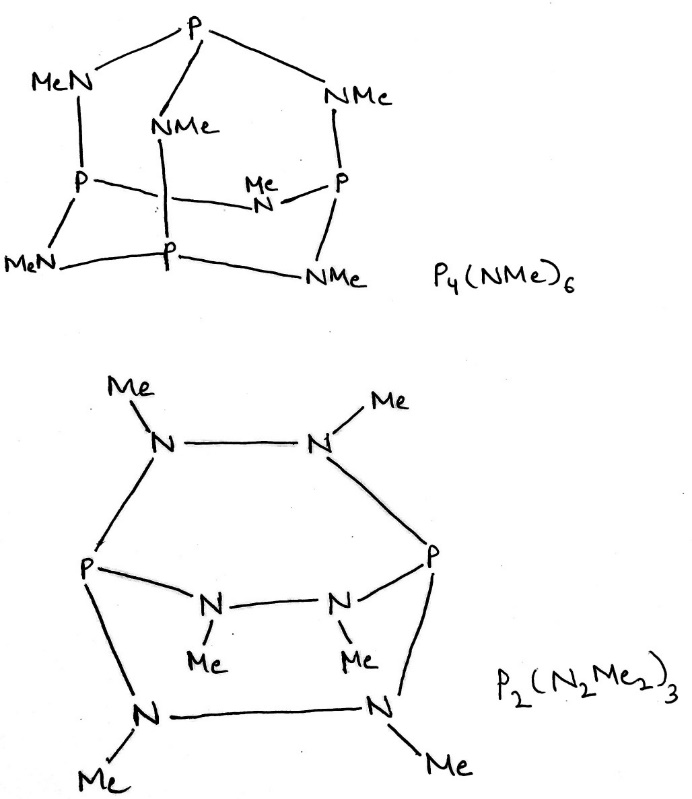
The range of PN compounds is extensive, and includes cyclic and polymeric phosphazenes,
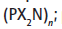
phosphazenes form highly flexible elastomers.
Phosphazenes refer to classes of organophosphorus compounds featuring phosphorus(V) with a double bond between P and N.
Polyphosphazenes include a wide range of hybrid inorganic-organic polymers with a number of different skeletal architectures with the backbone P-N-P-N-P-N-.In nearly all of these materials two organic side groups are attached to each phosphorus center. Linear polymers have the formula (N=PR1R2) n, where R1 and R2 are organic. Other architectures are cyclolinear and cyclomatrix polymers in which small phosphazene rings are connected together by organic chain units. Other architectures are available, such as block copolymer, star, dendritic, or comb-type structures. More than 700 different polyphosphazenes are known, with different side groups (R) and different molecular architectures.