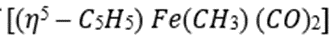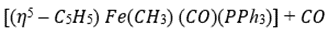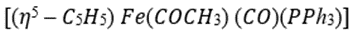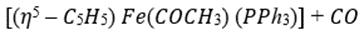Correct option is C
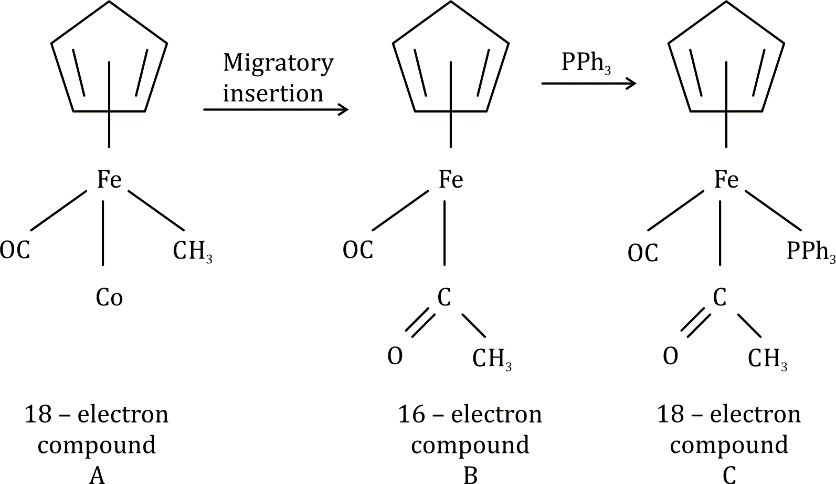
Two ligands can also react together to produce a new complex that still has the composite ligand attached to the metal ready for further modification. This process involves migration of one of the ligands from the metal to the other ligand and insertion of one of the ligands into the other metal–ligand bond and is known as migratory insertion. A cis arrangement of the ligands is required and the migrating group (X) retains its stereochemistry (if any) during the migration. The insertion process is reversible and, as the metal effectively loses a ligand in the process, the overall insertion may be driven by the addition of extra external ligands (L) to produce a coordinatively saturated complex. The group to be inserted must be unsaturated in order to accommodate the additional bonds and common examples include carbon monoxide, alkenes, and alkynes producing metal–acyl, metal–alkyl, and metal–alkenyl complexes, respectively.
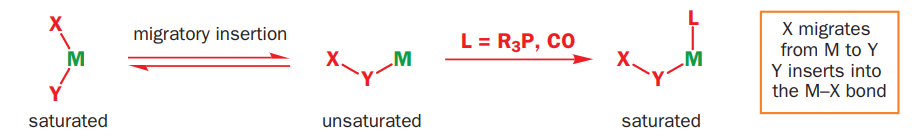
Cyclopentadienyl ligand
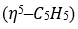
donates five electrons, Fe [[Ar]3d⁶4s²] donates eight electrons, both CO and PPh3 are two electron donors, COCH3 is a one-electron donor and CH3 is also a one-electron donor.