Correct option is B
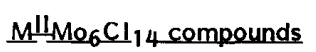
The structural formula is
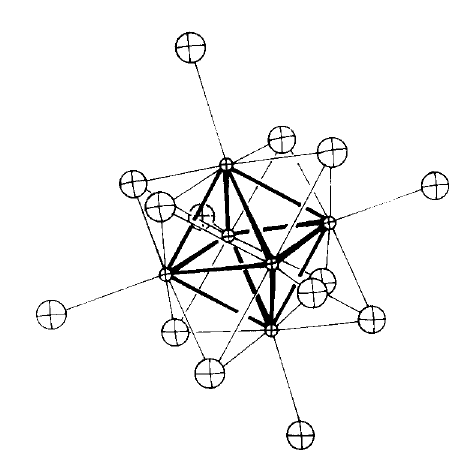
The Mo-Mo and Mo-Cl distances are about 2.60 and 2.47Å respectively. The six apical, terminal, Cl ligands lead to a quasi undeformed octahedral site (point group symmetry D3d) occupied by the M2+ cation.
When M is monovalent, two cations are to be inserted in the structure in order to counterbalance an
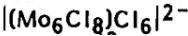
anion. Such compounds have been obtained for Li, K, Cs and Cu.
These ternary molybdenum +II chlorides exhibit a diamagnetic behavior except in the case of 3d (V, Cr, Mn, Fe, Co, Ni) or Eu and Yb element compounds which show M2+ion paramagnetism (high spin configuration for 3d-cation compounds) : effectively, from molybdenum electronic state and anionic and cationic charge transfers, one can derive a number of 24 valency electrons per cluster corresponding to twelve two-electrons Mo-Mo bonds directed alongside the edges of the Mo6 octahedra.
6 Mo ([Kr] 4d⁵5s¹)=6×6=36
Chloride as a triply bridging (μ3) ligand donates five electrons=8×5=40
Chloride as a terminal ligand donates one electron=6×1=6
Total valence electrons=82







