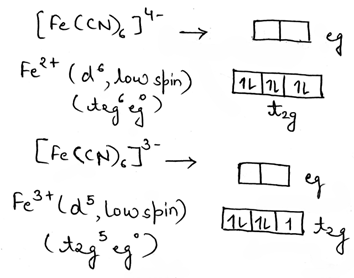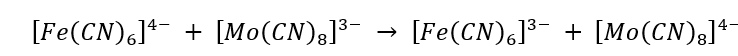Correct option is D
Outer sphere refers to an electron transfer (ET) event that occurs between chemical species that remain separate and intact before, during, and after the ET event. Both the reactants (i.e., oxidant and reductant) should be kinetically inert. The electron transfer is very fast when both the complexes are low spin including that the electron transfer takes place from t2g (π*) of reductant to the t2g (π*) of oxidant. The rates of electron transfer are much faster between the complexes which have π-acceptor ligands (like CN-, phen, bpy etc). The π-acceptor ligands have vacant π* orbitals that can accept electron being transferred, then pass them on to the receiving metal ion (i.e., oxidant). Outer sphere electron transfer reactions between complexes of different metal cations i.e., cross reactions are faster than self-exchange reactions.
In the given reaction, electron transfer is spin allowed. The electron transfer occurs from t2g of [Fe (CN)6]4- to t2g of [Fe (CN)6]3- complex. The ligand CN- is unsaturated and a π-acceptor which facilitates the electron tunneling.