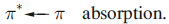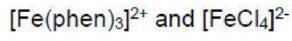Correct option is B
In metal complexes, intense absorptions (typically in the UV or visible part of the electronic spectrum) may arise from ligand-centred
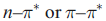
transitions, or from the transfer of electronic charge between ligand and metal orbitals. The latter fall into two categories:
transfer of an electron from an orbital with primarily ligand character to one with primarily metal character (ligand-to-metal charge transfer, LMCT).
transfer of an electron from an orbital with primarily metal character to one with primarily ligand character (metal-to-ligand charge transfer, MLCT).
LMCT occurs when a ligand that is easily oxidized is bound to a metal centre (usually one in a high oxidation state) that is readily reduced. Ligand-to-metal charge transfer may give rise to absorptions in the UV or visible region of the electronic spectrum. The LMCT band moves to lower wavelength (higher energy) as the metal centre becomes harder to reduce. The values of the absorption maxima for
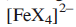
with different halidoligands illustrate a shift to longer wavelength (lower energy) as the ligand becomes easier to oxidize (I- easier than Br-, easier than Cl-).
LMCT transition occurs in
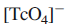
This transition corresponds to the promotion of an electron from an orbital that is mainly oxygen lone pair in character to a low-lying, mainly Tc-centred orbital.
Metal-to-ligand charge transfer typically occurs when the ligand has a vacant, low-lying π* orbital, for example, CO, py, bpy, phen and other heterocyclic, aromatic ligands. Often, the associated absorption occurs in the UV region of the spectrum and is not responsible for producing intensely coloured species. In addition, for ligands where a ligand-centred
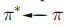
transition is possible (e.g. heterocyclic aromatics such as bpy), the MLCT band may be obscured by the