Correct option is A
The most fundamental reaction a complex can undergo is ligand substitution, a reaction in which one Lewis base displaces another from a Lewis acid:
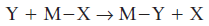
This class of reaction includes complex formation reactions, in which the leaving group, the displaced base X, is a solvent molecule and the entering group, the displacing base Y, is some other ligand.
The rate at which one complex converts into another is governed by the height of the activation energy barrier that lies between them. Thermodynamically unstable complexes that survive for long periods (by convention, at least a minute) are commonly called ‘inert’. Complexes that undergo more rapid equilibration are called labile.
Nonlability is common among the complexes of the 4d and 5d series, which reflects the high ligand field stabilization energy and strength of the metal-ligand bonding.
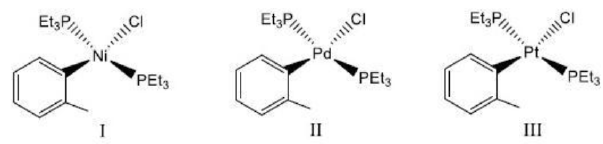
Nickel belongs to 3d series. Palladium belongs to 4d series. Platinum belongs to 5d series.
The greatest number of square planar complexes usually occur with transition metal ions of d8 electronic configuration. These include complexes of Ni(II), Pd(II), Pt(II), Rh(I), Ir(I) and Au(III). Square planar complexes of Pt (II) have been extensively studied and analysed because : (i) square planar complexes of Pt(II) are most stable, (ii) easy to synthesize, (iii) Pt (II) complexes of C.N. 4 are always square planar unlike Ni (II) complexes which form tetrahedral complexes with weak ligands and square planar complexes with strong ligands, (iv) substitution reactions in Pt(II) complexes undergo at slow rates and are, therefore, convenient for laboratory study. Ligand substitution reactions in Ni(II) and Pd(II) square planar complexes undergo 106 and 107times more rapidly than Pt(II) complexes respectively.