Correct option is A
In the model of an octahedral complex used in crystal-field theory, six-point negative charges representing the ligands are placed in an octahedral array around the central metal ion. These charges (which we shall refer to as the ‘ligands’) interact strongly with the central metal ion, and the stability of the complex stems in large part from this attractive interaction between opposite charges. However, there is a much smaller but very important secondary effect arising from the fact that electrons in different d orbitals interact with the ligands to different extents.
Electrons in dz2 and dx2 -y2 orbitals are concentrated close to the ligands, along the axes, whereas electrons in dxy, dyz, and dzx orbitals are concentrated in regions that lie between the ligands. As a result, the former are repelled more strongly by the negative charge on the ligands than the latter and lie at a higher energy. Group theory shows that the two eg orbitals have the same energy, and that the three t2g orbitals also have the same energy. This simple model leads to an energy-level diagram in which the three degenerate t2g orbitals lie below the two degenerate eg orbitals. The separation of the two sets of orbitals is called the ligand-field splitting parameter, Δo (where the subscript O signifies an octahedral crystal field).
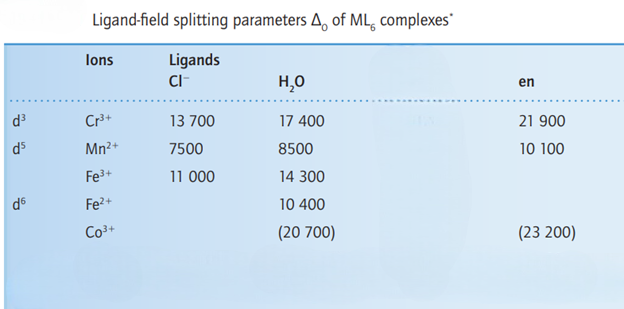
The ligand-field splitting parameter, Δo , varies systematically with the identity of the ligand. The energy of the lowest energy electronic transition (and therefore Δo ) increases as the ligands are varied along the series. The same order is followed regardless of the identity of the metal ion. Thus, ligands can be arranged in a spectrochemical series, in which the members are arranged in order of increasing energy of transitions that occur when they are present in a complex:
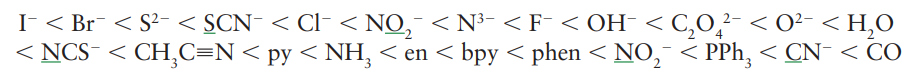
Thus, the series indicates that, for the same metal, the optical absorption of the cyano complex will occur at higher energy than that of the corresponding chlorido complex. A ligand that gives rise to a high-energy transition (such as CO) is referred to as a strong-field ligand, whereas one that gives rise to a low-energy transition (such as Br-) is referred to as a weak-field ligand.
The oxidation state of the metal also contributes to the size of Δo between the high and low energy levels. As the oxidation state increases for a given metal, the magnitude of Δo increases. The smaller distance between the ligand and the metal ion results in a larger Δo , because the ligand and metal electrons are closer together and therefore repel more.
More is the nuclear charge; more will be the repulsion between the d orbital (having electrons) of the metal and the electrons of the ligand. The higher oxidation state of the metal causes the ligands to approach more closely to it, and therefore the ligands cause more splitting of the metal d orbitals.
In [TiF6]3-, Ti is in +3 oxidation state. This complex comprises of weak field ligands.
In [MnF6]2-, Mn is in +4 oxidation state. This complex comprises of weak field ligands.
In [Co(en)3]3+, Co is in +3 oxidation state. This complex comprises of strong field ligands.
In [Fe (H2O)6]2+ , Fe is in +2 oxidation state.