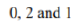Correct option is B
The cyclopentadienyl metallocenes of the elements of transition metals of 3d series are isomorphous. Except ferrocene, all other metallocenes are paramagnetic.
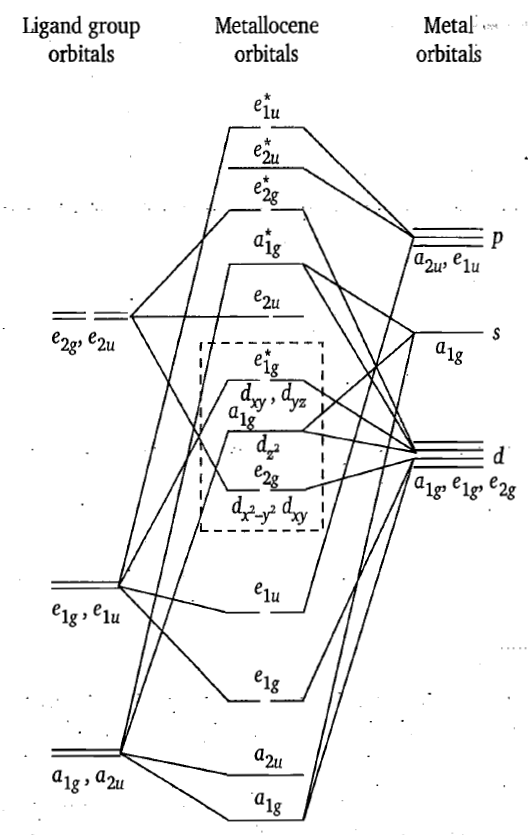
The molecular orbital energy diagram supported by photoelectron spectroscopy studies in conjunction with ligand field theory for ferrocene in figure.
The energy of a1g orbital of cyclopentadiene is too much less than a1g (4s or 3dz2 ) orbitals, therefore, a1g orbital of the ring interacts slightly with a1g orbitals of iron. Thus, a1g bonding molecular orbital is mainly ligand based and a1g (3dz2) orbital is non-bonding and localized on the metal. Similarly, the a2u orbital has little interaction with even higher energy Fe 4pz orbital with which it is formally able to combine. The pair of e1g MOs arises from the bonding combination of the ligand e1g orbitals with e1g (dxz and dyz) orbitals on metal and form two strong pi-bonds. This is due to the reason that energy difference between e1g orbitals of ligand and e1g orbitals of metal and their symmetries are well-matched. This is the main contribution to the stability of the complex. The corresponding anti-bonding e1g* orbitals are unoccupied but involve in electronic transitions. Although the interaction of ligand e1u orbitals with metal e1u (px, py) orbitals is favourable, but due to large energy difference they interact very little and the e1u molecular orbitals are again mainly ligand based but with some contribution from metal 4px and 4py orbitals. Since the energy of ligand e2g orbitals is higher than those of e2g (dxy and dx2-y2) orbitals of same symmetry of the metal, they do not interact very effectively and, therefore, the e2g molecular orbitals are metal based and are non-bonding. Also the metal a1g (i.e., dz2) orbital does not interact very effectively with a1g orbitals of the ligand, therefore, is localized on metal and is non-bonding.
The orbitals of metallocenes that are of most interested are those having the greatest d-orbital character. These orbitals are the highest occupied and the lowest unoccupied molecular orbitals (HOMO and LUMO respectively).
The electron-electron repulsion between the pi-electron density of the ligand e2g orbitals and the metal e2g (dx2-y2 and dxy) orbitals is minimum as the lobes of the dx2-y2 and dxy orbitals are not directed towards the cyclopentadiene ring. The a1g (dz2) orbital has one lobe and e1g (dyz and dzx) orbitals have two lobes directed towards the cyclopentadiene ring and, therefore, repulsion between a1g of metal and that of Cp ring lower than between e1g of metal and e1g of Cp ring.
Therefore, the order of energy of molecular orbitals
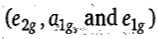
which are localized on metal is
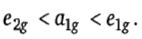
It can be seen from molecular orbital energy level diagram that ferrocene contains 18 valence electrons in bonding and non-bonding MOs and the anti-bonding MOs are unoccupied. Since the bonding MOs are localized on Cp rings, therefore, 12 electrons supplied by two Cp rings (2x6e-) are filled in bonding MOs
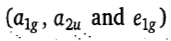
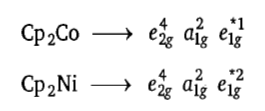
and 6 electrons supplied by Fe2+ ion are filled in non-bonding e2g and a1g orbitals. Thus, the distribution of six Fe2+ion electrons in frontier molecular orbital is
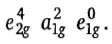
. The a1g is HOMO and e1g is LUMO in ferrocene. Ferrocene obeys the 18-electron rule. According to ligand field theory, the energy difference between e2g and a1g MOs is less than the pairing energy, therefore, these orbitals are first singly filled and then pairing occurs.