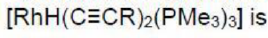Correct option is B
The isomers in which the same types and number of ligands are coordinated to the metal atom or cation but with different spatial arrangements are called stereoisomers. In other words, stereoisomers are isomers that differ only in the spatial arrangement of ligands coordinated to metal cation or atom. Stereoisomerism is classified into two types :
(1) Geometrical isomerism
(2) Optical isomerism
Geometrical isomerism
Stereoisomers in which the relative positions or orientations of the ligands or more specifically donor atoms around the metal cation is different are called geometrical isomers and this phenomenon is called geometrical isomerism. Geometrical isomers can not be inter-converted without breaking of M-L bonds. Geometrical isomers exists only in pairs, in one isomer the two particular ligands are adjacent to each other and in the other the two are in opposite sides in the structural formula. Thus, the isomer in which two particular ligands (either identical or non-identical) occupy the adjacent positions of each other is called cis-isomer and the isomer in which two particular ligands occupy opposite positions to each other is called trans-isomer (The Latin word cis means next to, trans means across). cis- and trans- isomers are different compounds with different properties like melting points, dipole moments, solubility, colours and chemical properties. Geometrical isomerism is most common in complexes having coordination number of 4 and 6.
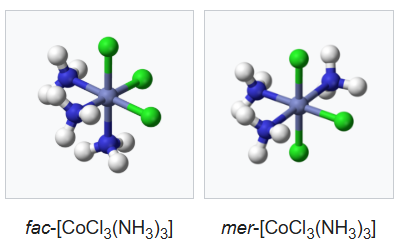
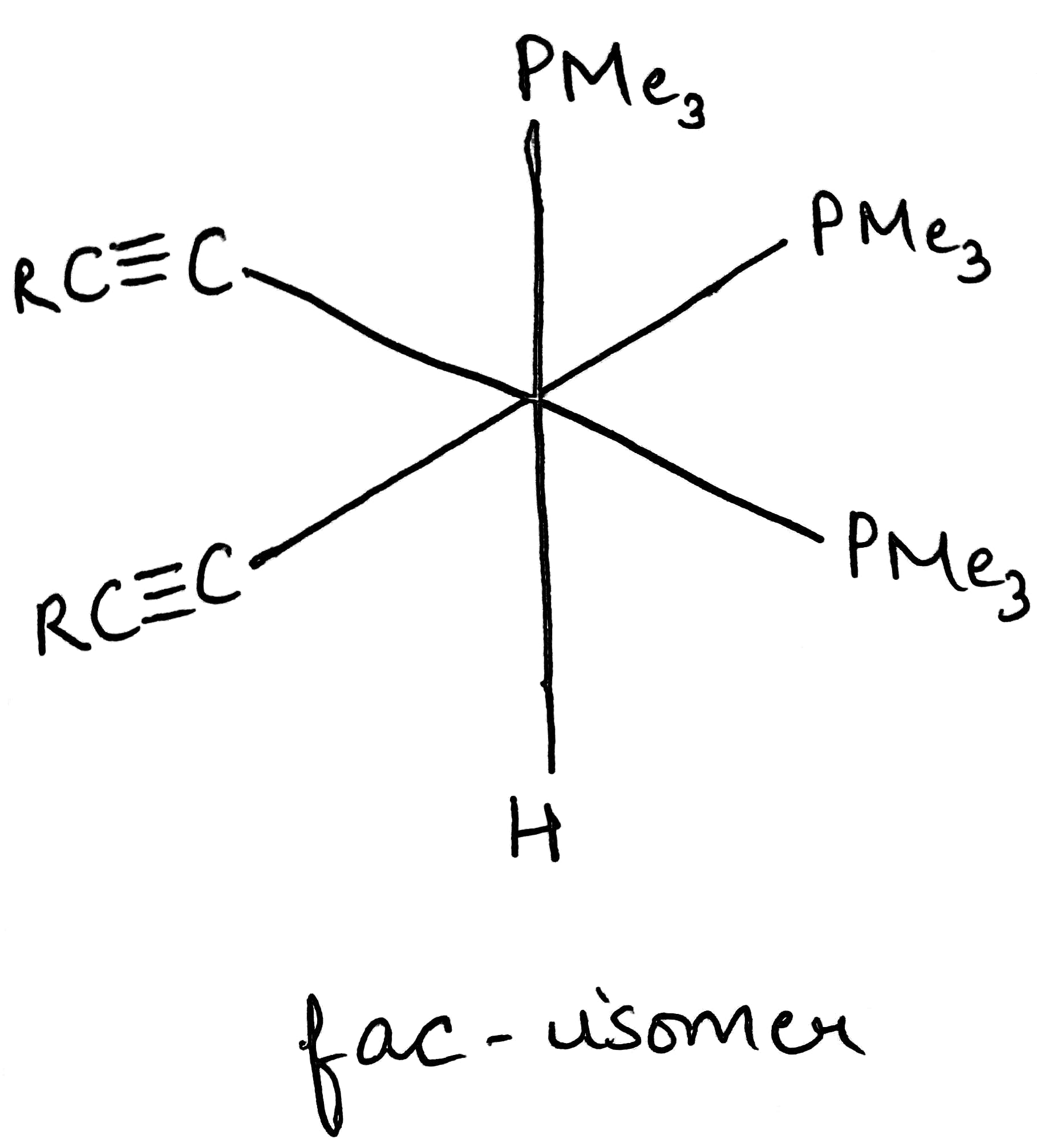
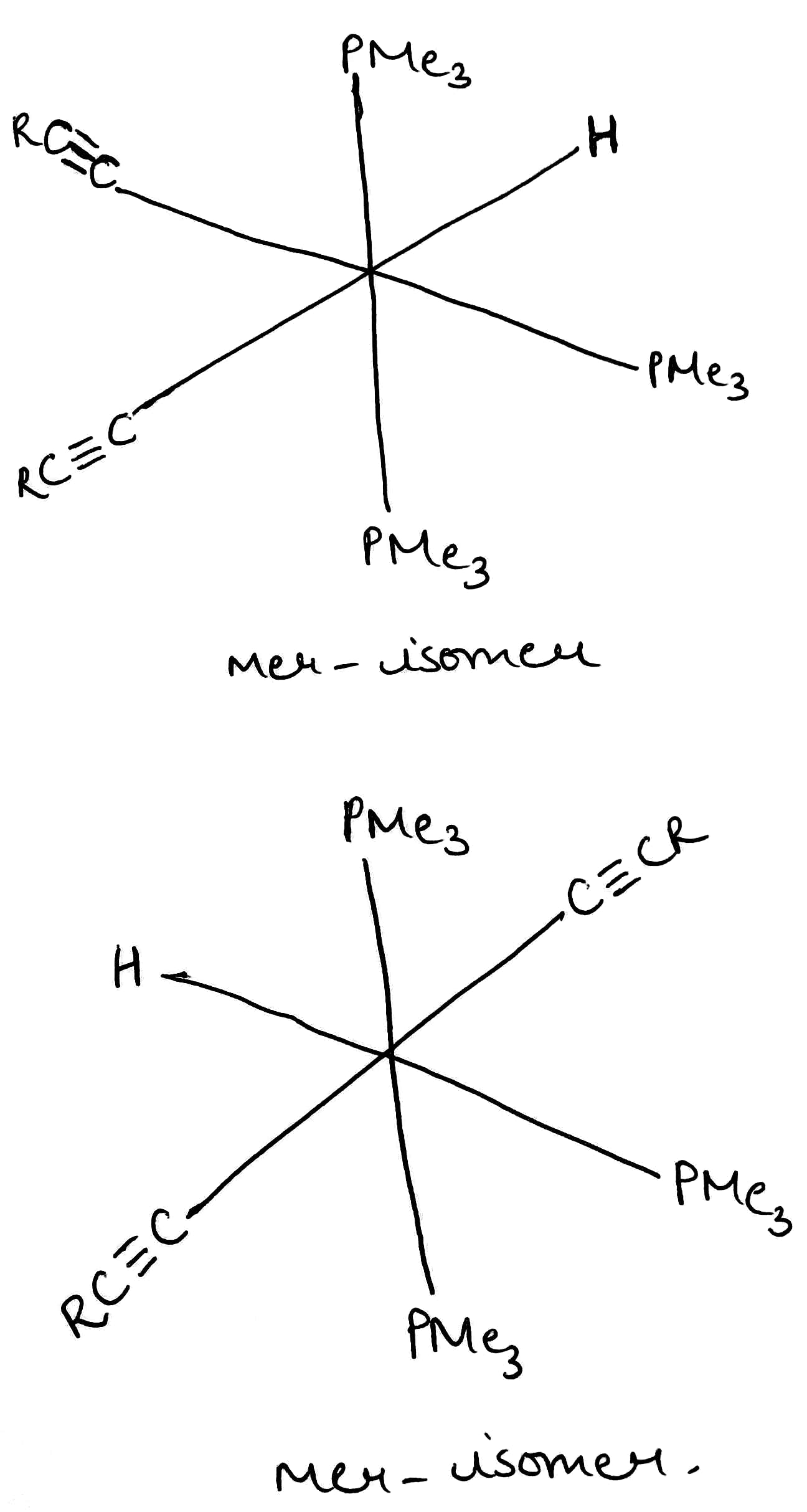
A type of isomerism in octahedral MX3Y3 complexes is facial–meridional (or fac–mer) isomerism, in which different numbers of ligands are cis or trans to each other. When three identical ligands occupy one face of an octahedron, the isomer is said to be facial, or fac. In a fac isomer, any two identical ligands are adjacent or cis to each other. If these three ligands and the metal ion are in one plane, the isomer is said to be meridional, or mer. A mer isomer can be considered as a combination of a trans and a cis, since it contains both trans and cis pairs of identical ligands.