Correct option is D
Electron paramagnetic resonance (EPR) spectroscopy (also called electron spin resonance (ESR) spectroscopy), is used to study paramagnetic species with one or more unpaired electrons, e.g. free radicals, diradicals, metal complexes containing paramagnetic metal centres, defects in semiconductors and irradiation effects in solids. While diamagnetic materials are EPR silent, paramagnetic species always exhibit an EPR spectrum. This consists of one or more lines, depending on the interactions between the unpaired electron (which acts as a ‘probe’) and the molecular framework in which it is located. Analysis of the shape of the EPR spectrum (the number and positions of EPR lines, their intensities and line widths) provides information about the paramagnetic species, e.g. the structure of a free radical, characterization of the coordination sphere around the metal centre in a coordination complex, or the presence of multiple paramagnetic species.
EPR spectroscopic measurements can be performed at high, room or low (4 K) temperature. Samples may be solid (single crystal or powder) or liquid (fluid or frozen solution, a ‘glass’).
The Zeeman electronic effect
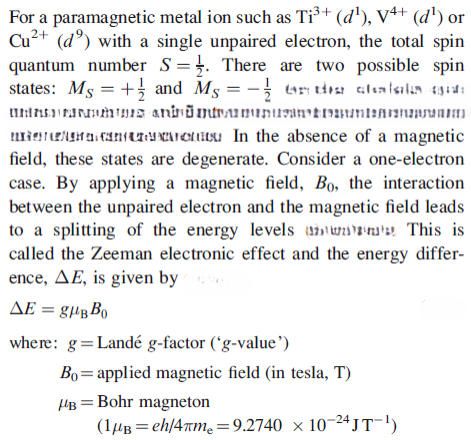

electromagnetic spectrum. Thus, by supplying appropriate microwave radiation to the sample, electron spin transitions between the two energy states occur. The system is then in resonance, and the recording of these transitions represents the EPR spectrum. Usually, an EPR spectrometer operates at a constant microwave frequency (measured in gigahertz, GHz) and the magnetic field (measured in gauss or tesla,
is varied until the energy separation of the two spin states coincides with the microwave radiation energy. Standard EPR spectrometers operate at 9–10 GHz (so-called ‘X-band’), but there are also domains of lower and higher microwave frequencies: 1–2GHz (L-band), 2–4 GHz (S-band), 35 GHz (Q-band) and 95 GHz (W-band). Recently developed FT-EPR spectrometers (as opposed to continuous wave instruments) produce increased spectral resolution and their use has widened the scope of systems that can be investigated (e.g. the second coordination sphere around a paramagnetic metal centre in a metalloprotein).
EPR Spectra
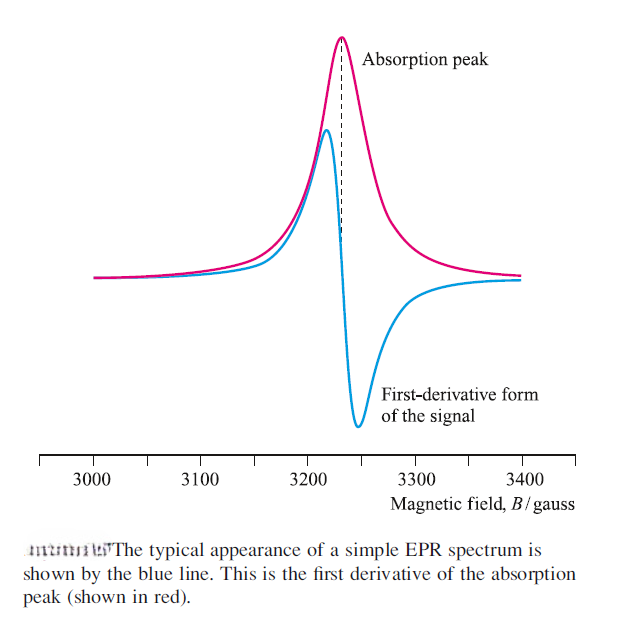
The form in which an EPR spectrum is recorded is the first derivative of an absorption peak, because in this form the detection is more sensitive and the signal : noise ratio is improved due to intrinsic electronic properties resulting from modulation of the magnetic field. The point at which the derivative curve is zero (i.e. crosses the baseline) corresponds to the absorption maximum, and the magnetic field, Bsample , at this
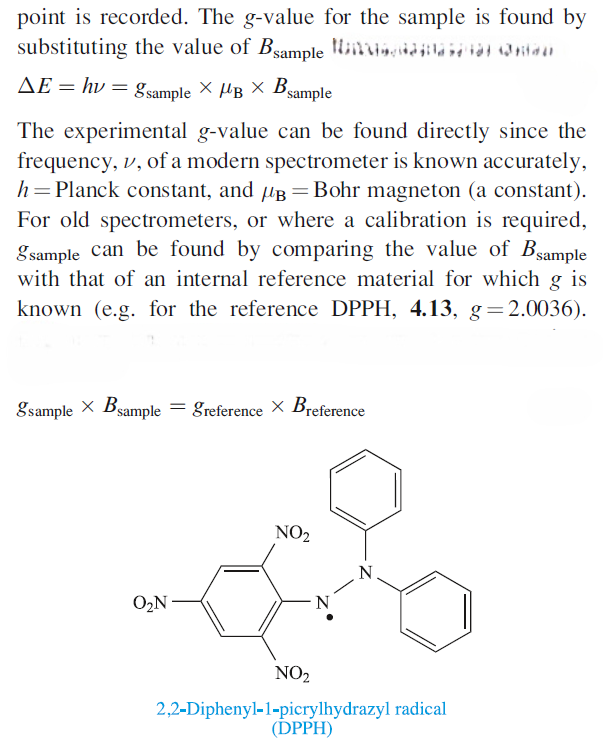
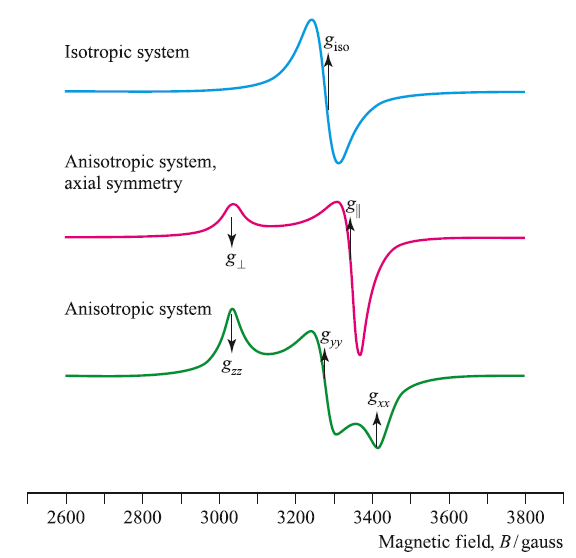
The g-value obtained from an EPR experiment provides diagnostic information about the system being investigated. For a paramagnetic metal centre, the g-value is characteristic of the oxidation state (i.e. the number of unpaired electrons), the coordination environment and the molecular symmetry. However, unless a system has cubic symmetry (i.e. it belongs to the Td, Oh or Ih point groups and is isotropic), the g-value depends on the orientation of the molecular principal axis with respect to the magnetic field. Such systems are said to be anisotropic. By rotation of the sample placed in the magnetic field in three orthogonal planes, three g-values are therefore obtained. Each g-value is associated with one of the three orthogonal axes. Three cases must now be considered:

In addition to the information available from g-values, we can obtain information about nuclei with nuclear spin quantum number
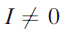
which are close to the paramagnetic centre. The spins of such nuclei interact magnetically with the unpaired electron and give rise to a hyperfine interaction. There is a direct analogy here with coupling of nuclear spins in NMR spectroscopy. The hyperfine interaction is added to the Zeeman electronic interaction, leading to a further splitting of the energy levels.

The EPR spectrum of one line (as for a scalar gyromagnetic factor) is split and the number of lines in the hyperfine pattern is given by 2nI+1, where n is the number of equivalent nuclei with spin quantum number I.
According to question,
Number of unpaired electrons=3
n=1 (only one metal ion is present)
I=7/2
Number of lines=3×(2×1×(7/2)+1)=24











