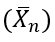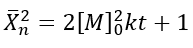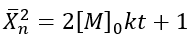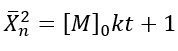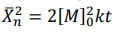Correct option is A
The number-average degree of polymerization (Xn) describes the average number of monomer units in a polymer chain. In the case of a self-catalyzed polyesterification reaction, which is a third-order reaction, the degree of polymerization can be derived based on the reaction kinetics and the initial monomer concentration.
Polymerization Kinetics:
• For a third-order reaction, the rate of reaction is proportional to the cube of the concentration of the monomer.
• The number-average degree of polymerization (Xn) increases as the monomers react to form polymer chains, and it depends on the initial monomer concentration ([M]o) and the rate constant (k).