Correct option is A
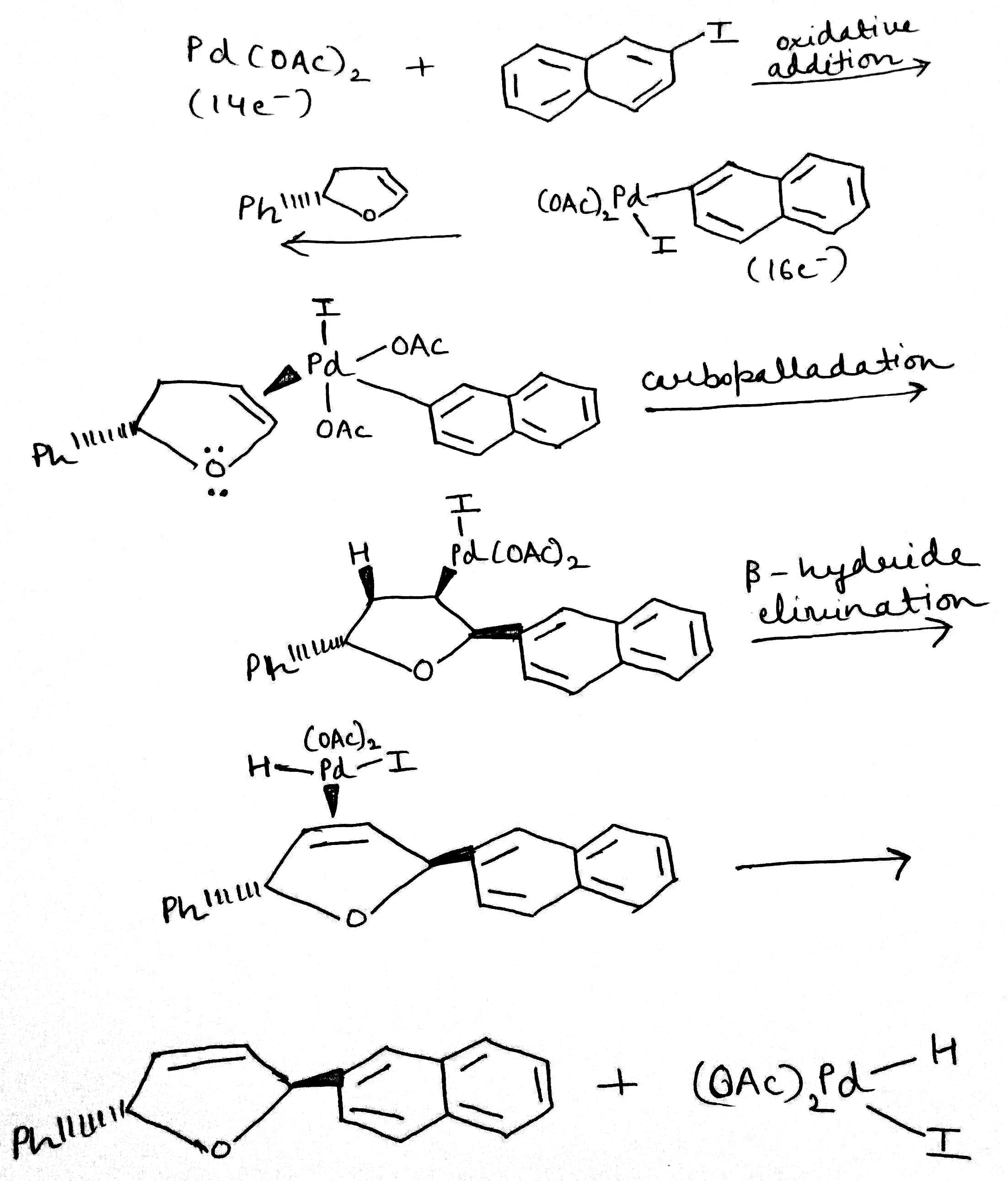
The palladium-catalysed addition of aryl, vinyl, or substituted vinyl groups to organic halides, the Heck reaction, is one of the most synthetically useful palladium-catalysed reactions. The mechanism involves the oxidative addition of the halide, insertion of the olefin, and elimination of the product by a β-hydride elimination process. A base then regenerates the palladium (0) catalyst. The whole process is a catalytic cycle.
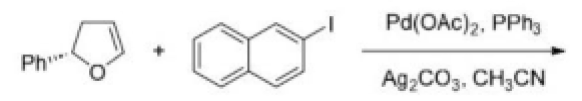
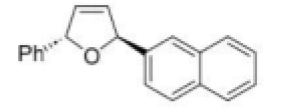
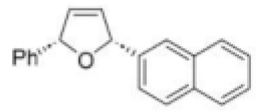
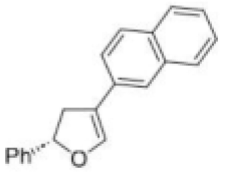
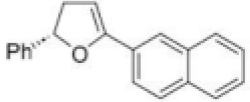
The given Heck reaction involves a naphthyl iodide (Ar2= 2-naphthyl). However, the enol ether has two diastereotopic faces: syn or anti to the aromatic substituent (Ar1=Ph). Palladium is very sensitive to steric effects and generally forms less hindered complexes where possible. Thus, coordination of the palladium (II) intermediate occurs on the face of the enol ether anti to Ar1.This in turn controls all the subsequent steps, which must be syn, leading to the trans product. The requirement for syn β-hydride elimination also explains the regiochemical preference of the elimination. In this cyclic structure there is only one hydrogen (green) that is syn; the one on the carbon bearing the naphthyl substituent is anti to the palladium and cannot be eliminated.
Information booster
Oxidative addition is a process that increases both the oxidation state and coordination number of a metal centre.
A carbometallation is any reaction where a carbon-metal bond reacts with a carbon-carbon π-bond to produce a new carbon-carbon σ-bond and a carbon-metal σ-bond
β-Hydride elimination is a reaction in which an alkyl group bonded to a metal centre is converted into the corresponding metal-bonded hydride and an alkene. The alkyl must have hydrogens on the β-carbon. In the β-hydride elimination step, the palladium and hydride must be coplanar for reaction to take place, as this is a syn elimination process.
Reductive elimination is an elementary step in organometallic chemistry in which the oxidation state of the metal center decreases while forming a new covalent bond between two ligands.