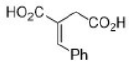
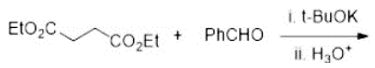Correct option is A
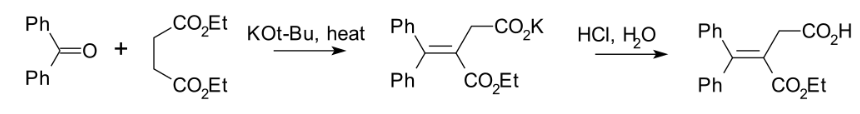
The Stobbe condensation entails the reaction of an aldehyde or ketone with an ester of succinic acid to generate alkylidene succinic acid or related derivatives. The reaction consumes one equivalent of metal alkoxide. Commonly, diethylsuccinate is a component of the reaction. The usual product is salt of the half-ester. An example is the reaction of benzophenone with diethyl succinate:
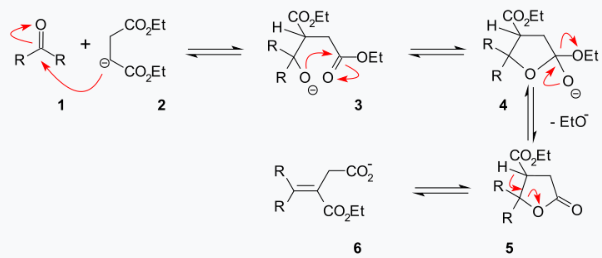
A reaction mechanism that explains the formation of both an ester group and a carboxylic acid group is centered on a lactone intermediate (5):
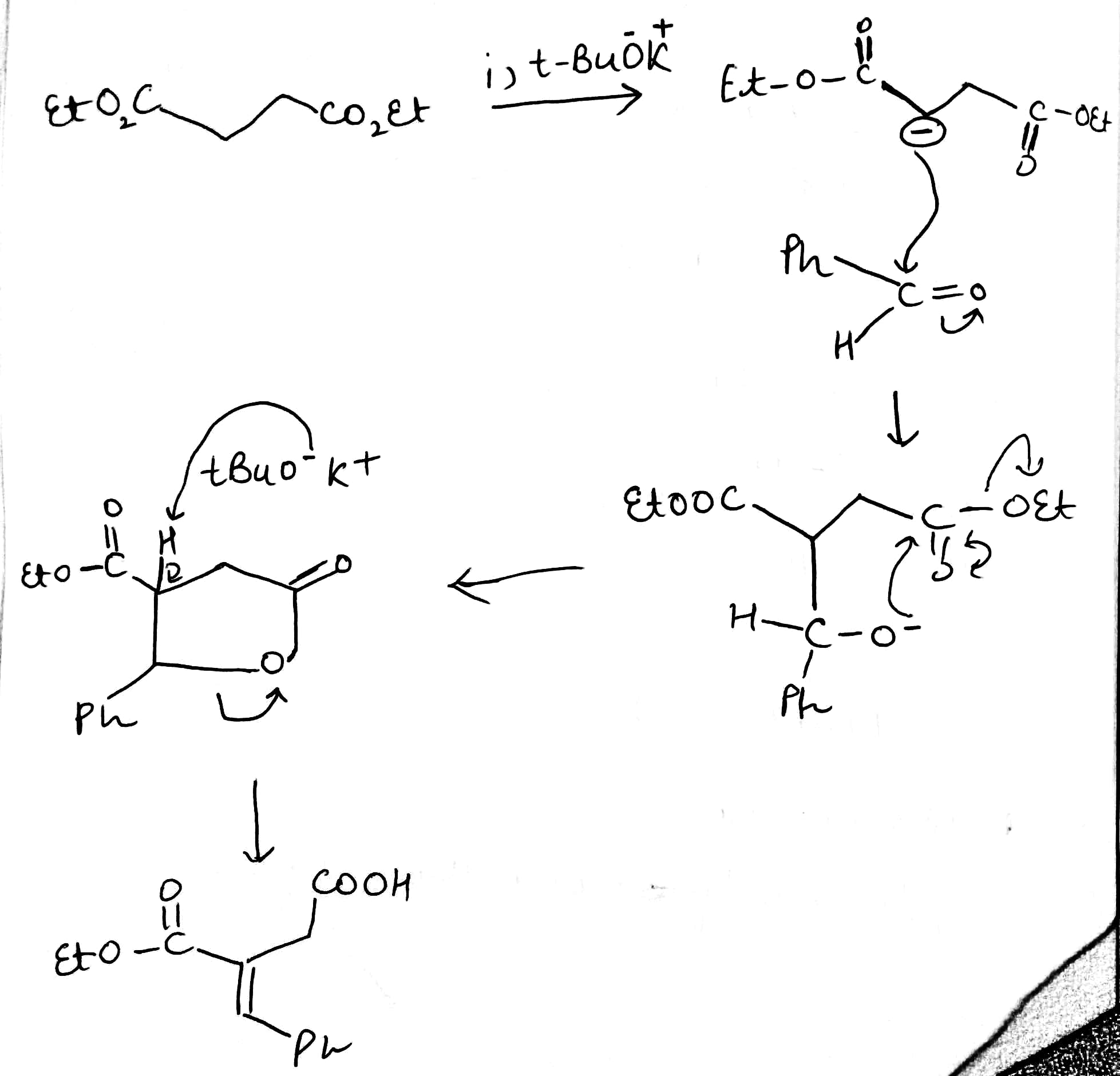
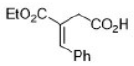
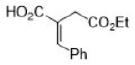
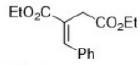
Ph group will be trans to the bulky -COOEt group.