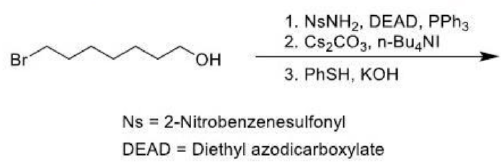
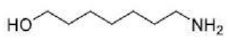
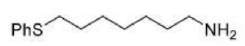
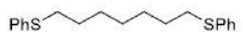
Correct option is D
The Mitsunobu reaction is an organic reaction that converts an alcohol into a variety of functional groups, such as an ester, using triphenylphosphine and an azodicarboxylate such as diethyl azodicarboxylate (DEAD) or diisopropyl azodicarboxylate (DIAD). In a typical protocol, one dissolves the alcohol, the carboxylic acid, and triphenylphosphine in tetrahydrofuran or other suitable solvent (e.g. diethyl ether), cool to 0 °C using an ice-bath, slowly add the DEAD dissolved in THF, then stir at room temperature for several hours. The alcohol reacts with the phosphine to create a good leaving group then undergoes an inversion of stereochemistry in classic SN2 fashion as the nucleophile displaces it.
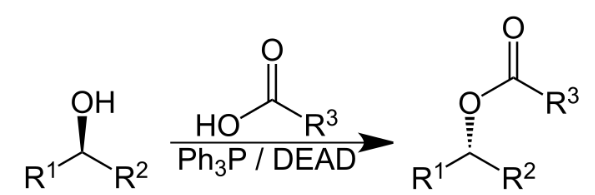
The reaction mechanism of the Mitsunobu reaction is fairly complex.
Initially, the triphenyl phosphine (2) makes a nucleophilic attack upon diethyl azodicarboxylate (1) producing a betaine intermediate 3, which deprotonates the carboxylic acid (4) to form the ion pair 5. The formation of the ion pair 5 is very fast.
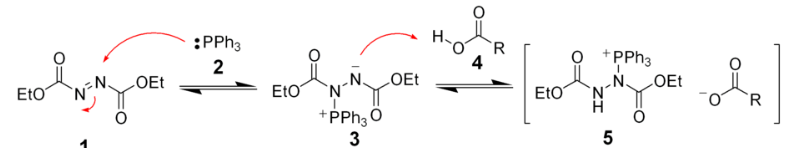
The second phase of the mechanism is proposed to be phosphorus-centered, the DEAD having been converted to the hydrazine.
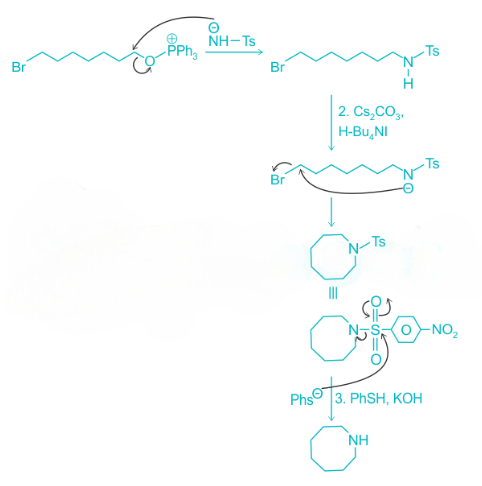
Cs2CO3 abstracts one acidic proton giving an anion. The resulting anion undergoes an intramolecular SN2 displacement reaction and gives a cyclized product. In the last step, PhSH acts as a nucleophile