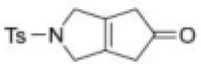
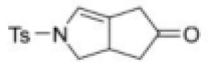
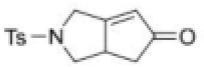
Correct option is D
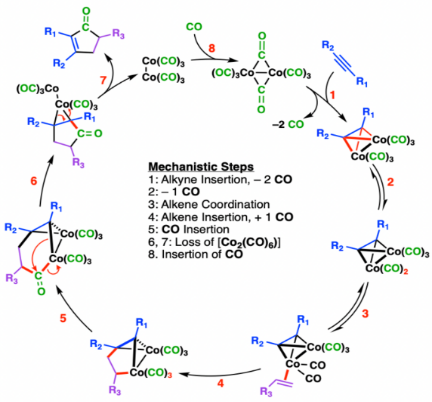
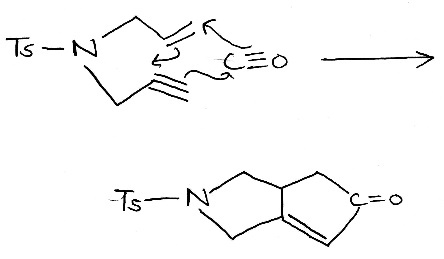
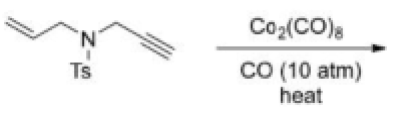
The Pauson–Khand reaction is a chemical reaction, described as a [2+2+1] cycloaddition. In it, an alkyne, an alkene, and carbon monoxide combine into a α,β-cyclopentenone in the presence of a metal-carbonyl catalyst.
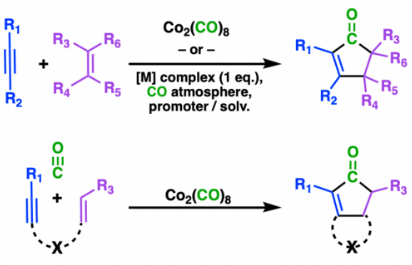
The reaction starts with dicobalt hexacarbonyl acetylene complex. Binding of an alkene gives a metallacyclopentene complex.CO then migratorily inserts into an M-C bond. Reductive elimination delivers the cyclopentenone. Typically, the dissociation of carbon monoxide from the organometallic complex is rate limiting.
1:Alkyne coordination, insertion and ligand dissociation to form an 18-electron complex;
2:Ligand dissociation to form a 16-electron complex;
3:Alkene coordination to form an 18-electron complex;
4:Alkene insertion and ligand association (synperiplanar, still 18 electrons);
5:CO migratory insertion;
6, 7:Reductive elimination of metal (loss of [Co2(CO)6]);
8:CO association, to regenerate the active organometallic complex