Correct option is B
β-Hydride elimination is a reaction in which an alkyl group bonded to a metal centre is converted into the corresponding metal-bonded hydride and an alkene. The alkyl must have hydrogens on the β-carbon. For instance butyl groups can undergo this reaction but methyl groups cannot. The metal complex must have an empty (or vacant) site cis to the alkyl group for this reaction to occur.
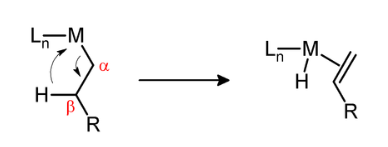
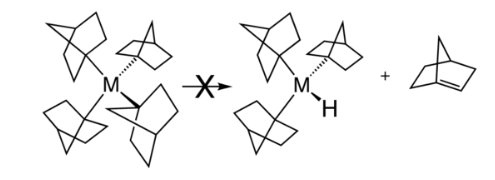
Several strategies exist for avoiding β-hydride elimination. The most common strategy is to employ alkyl ligands that do not have any hydrogen atoms at the β position. Common substituents include methyl and neopentyl. β-Hydride elimination is also inhibited when the reaction would produce a strained alkene. This situation is illustrated by the stability of metal complexes containing norbornyl ligands, where the β-hydride elimination product would violate Bredt's rule.
In organic chemistry, an anti-Bredt molecule is a bridged molecule with a double bond at the bridgehead. Bredt's rule is the empirical observation that such molecules only form in large ring systems. Bredt's rule results from geometric strain: a double bond at a bridgehead atom necessarily must be trans in at least one ring. For small rings (fewer than eight atoms), a trans alkene cannot be achieved without substantial ring and angle strain (the p orbitals are improperly aligned for a π bond).
Additional Knowledge
Agostic interaction refers to the intramolecular interaction of a coordinatively-unsaturated transition metal with an appropriately situated C-H bond on one of its ligands. The interaction is the result of two electrons involved in the C-H bond interaction with an empty d-orbital of the transition metal, resulting in a three-center two-electron bond. It is a special case of a C-H sigma complex.







