Correct option is C
Electron counting scheme for High Nuclearity Clusters
The HNCC are large, often structurally complex and most of them contain heavier (2nd and 3rd series) transition metals. These are not amenable to conventional interpretation of their electronic structures. Much effort has been devoted to find some relationship between their structure and the number of electrons available for cluster formation. All of these large clusters are regarded as electron deficient since there are insufficient electrons to permit the assignment of an electron pair bond between each adjacent pair of metal atoms.
The TEC in the cluster has the contribution from (i) the number of valence electrons of each metal, (ii) 2e for each CO group irrespective of its bonding, terminal or bridged, (iii) le for each negative charge and (d) number of valence electrons for each hetero atom and/ or interstitial atom, for example, 1 for H, 4 for C, 5 for N and so on. The polyhedral electron count (PEC) is calculated as follows:
PEC = TEC - 12e per skeletal metal atom
If a non-transition element is present on the vertex, then subtract 2e per element instead of 12e. Once we have the TEC and PEC, then, on the basis that n + 1 pairs of electrons are required for a polyhedron with n vertices (or alternatively, the number of vertices of the parent polyhedron is one less than the PEC), the most likely structure may be selected.


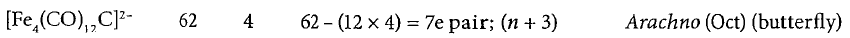
Fe ([Ar] 3d⁶4s²) contributes eight electrons.
C ([He] 2s²2p²) contributes four electrons.
Each CO ligand is a two electron donor.
Two negative charges gives two extra electrons.







