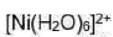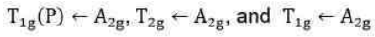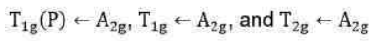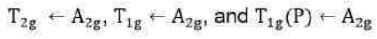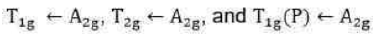Correct option is B
In d8 octahedral complexes, the d-orbitals split into t2g (lower energy) and eg (higher energy) orbitals. Thus, in ground state the electronic configuration is
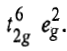
There is only one electronic arrangement for this electronic configuration
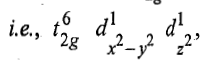
and the ground state term corresponding to this configuration is represented as
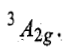
In a d8 octahedral complex two holes may be considered in eg orbitals as shown in figure.
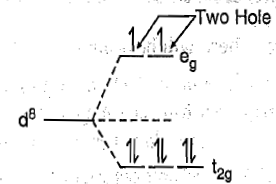
When an electron from any of the t2g orbitals is excited to any one of the eg orbitals, the electronic configuration becomes
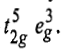
Now there will be two holes one in t2g and one in eg orbitals. Corresponding to this electronic configuration, there are two triply degenerate states. The lower energy state will arise when two holes occupy the orbitals as far apart as possible. This state is represented as
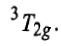
The holes may be present either in
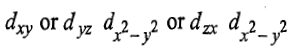
orbitals giving rise to lesser repulsions between two holes because two holes occupy more space in x, y and z-directions. Holes are effected in the opposite way as compared to electrons. There is another triply degenerate arrangement of two holes in which the two holes occupy the orbitals which are close together. The holes may be present in either
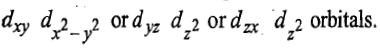
This is of higher energy and is represented as
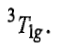
When both the electrons are excited, the electronic configuration becomes
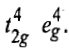
Now two holes are present in any of the two t2g orbitals either in
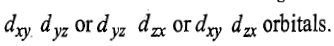
Therefore, this state is triply degenerate and is represented as
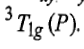
Since double excitation requires higher energy, therefore,
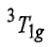
state is of highest energy state. There are four energy states including ground state. Thus, there are three transitions. The transitions of holes is similar to electronic transitions.

When ligand field strength increases, there will be bending of
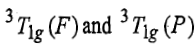
lines because these states are of same symmetry and there is inter electronic repulsion
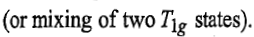
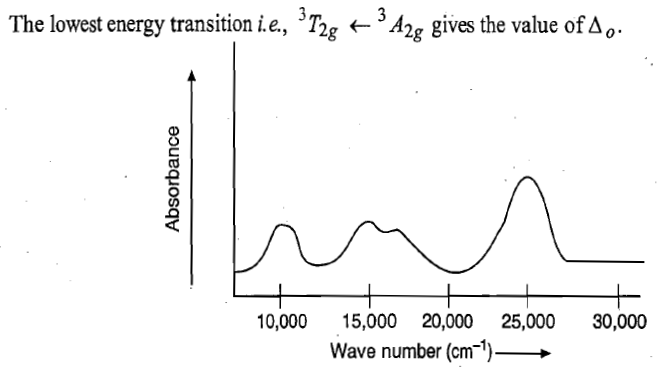

8700cm-1=1149nm
14500cm-1=689nm
25300cm-1=395nm