Correct option is C
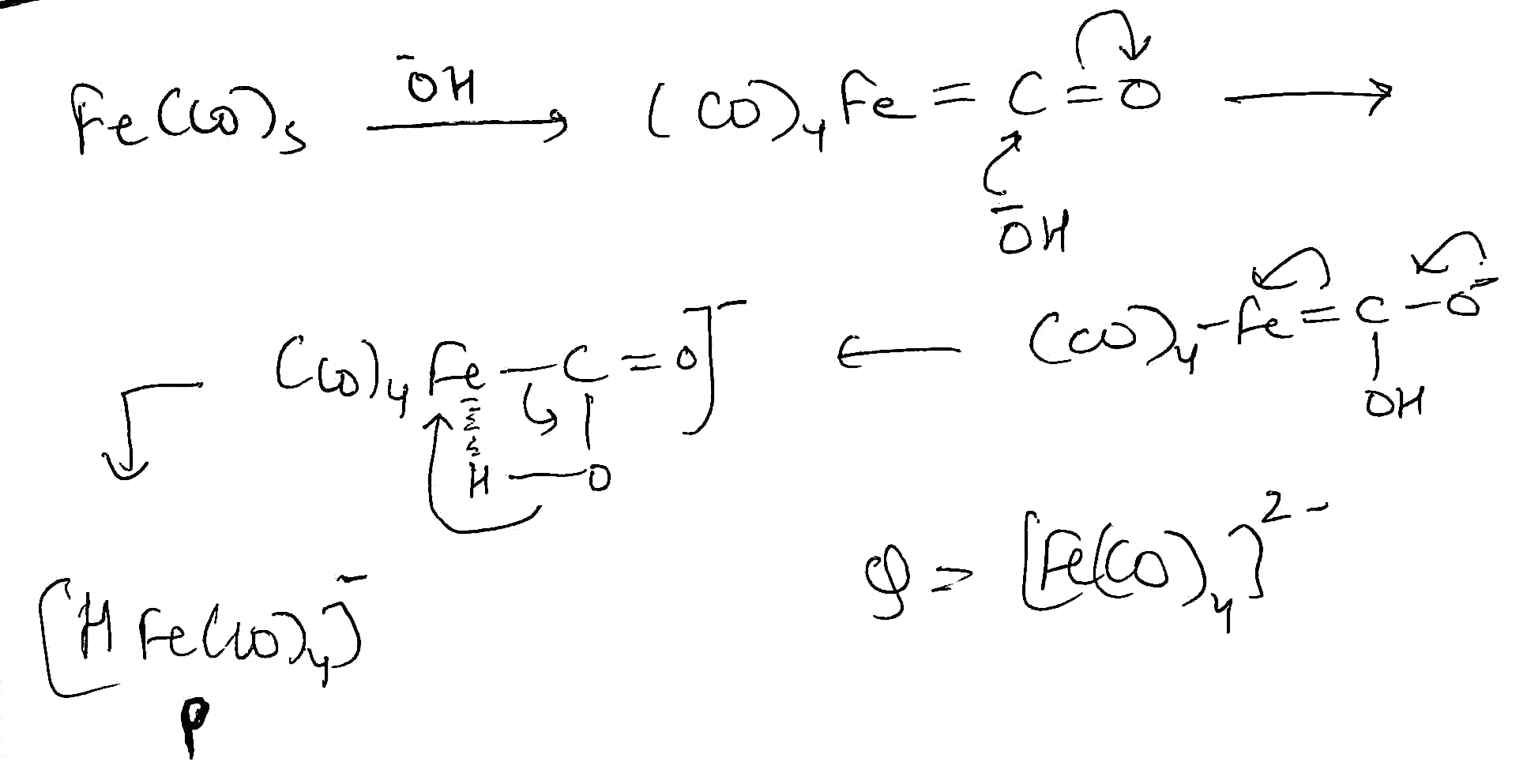
Option a:Carbon monoxide bonds to transition metals using "synergistic pi* back-bonding". The M–C bonding has three components, giving rise to a partial triple bond. A sigma (σ) bond arises from overlap of the nonbonding (or weakly anti-bonding) sp-hybridized electron pair on carbon with a blend of d-, s-, and p-orbitals on the metal. A pair of pi (π) bonds arises from overlap of filled d-orbitals on the metal with a pair of π*-antibonding orbitals projecting from the carbon atom of the CO. The latter kind of binding requires that the metal have d-electrons, and that the metal be in a relatively low oxidation state (0 or +1) which makes the back-donation of electron density favorable. As electrons from the metal fill the π-antibonding orbital of CO, they weaken the carbon–oxygen bond compared with free carbon monoxide, while the metal–carbon bond is strengthened. Because of the multiple bond character of the M–CO linkage, the distance between the metal and carbon atom is relatively short, often less than 1.8 Å, about 0.2 Å shorter than a metal–alkyl bond.
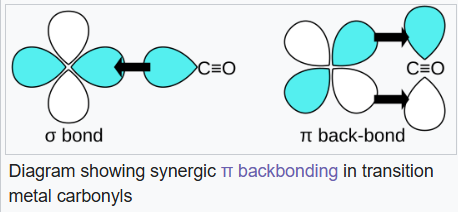
The energies of the νCO band for the metal carbonyls correlates with the strength of the carbon–oxygen bond, and inversely correlated with the strength of the π-backbonding between the metal and the carbon. The hexacarbonyls show decreasing π-backbonding as one increases (makes more positive) the charge on the metal. π-Basic ligands increase π-electron density at the metal, and improved backbonding reduces νCO.
Due to the presence of negative charge on the complex P, νCO (in P) is less than νCO in Fe(CO)5.
Option b:Formylation refers to any chemical processes in which a compound is functionalized with a formyl group (-CH=O). In organic chemistry, the term is most commonly used with regards to aromatic compounds (for example the conversion of benzene to benzaldehyde in the Gattermann–Koch reaction).
Formylation generally involves the use of formylation agents, reagents that give rise to the CHO group. Among the many formylation reagents, particularly important are formic acid and carbon monoxide. A formylation reaction in organic chemistry refers to organic reactions in which an organic compound is functionalized with a formyl group (-CH=O). The reaction is a route to aldehydes (C-CH=O), formamides (N-CH=O), and formate esters (O-CH=O).
Option c: The electronic configuration of Fe is [Ar] 3d⁶4s². CO is a two electron donor. Total electron count in [Fe(CO)4]2-is 8+(4×2)+2=18
Electronic configuration of Ni is [Ar] 3d⁸4s². Total electron count is Ni(CO)4 is 10+(4×2)=18
Option d: The first H2/CO mixture to be of commercial importance was obtained from the action of steam on red hot coke and, because of its origin, became known as water gas:
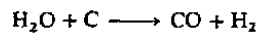
The ratio of hydrogen to carbon monoxide in water gas can be altered with the water gas shift reaction, which can be catalyzed by a variety of heterogeneous and homogeneous catalysts:

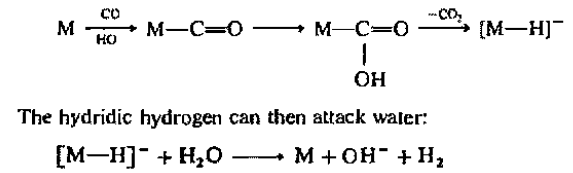
Commercially, the water gas shift reaction is usually carried out over Fe3O4. Metal carbonyl complexes such as
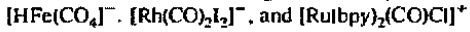
are effective and although all the mechanisms have not been worked out completely, the reactions may be viewed in general terms as beginning with a nucleophilic attack on coordinated carbon monoxide: