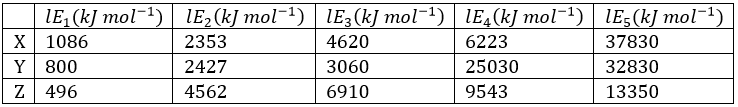
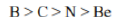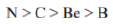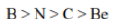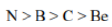Correct option is D
Ionization energy (IE) is the minimum energy required to remove the most loosely bound electron of an isolated gaseous atom, positive ion, or molecule. The first ionization energy is quantitatively expressed as
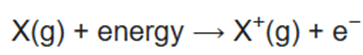
where X is any atom or molecule, X+ is the resultant ion when the original atom was stripped of a single electron, and e− is the removed electron. Roughly speaking, the closer the outermost electrons are to the nucleus of the atom, the higher the atom's ionization energy.
The nth ionization energy refers to the amount of energy required to remove the most loosely bound electron from the species having a positive charge of (n − 1). For example, the first three ionization energies are defined as follows:

Ionization energy generally increases from left to right within a given period (that is, row). As the nuclear charge of the nucleus increases across the period, the electrostatic attraction increases between electrons and protons, hence the atomic radius decreases, and the electron cloud comes closer to the nucleus because the electrons, especially the outermost one, are held more tightly by the higher effective nuclear charge.
After the removal of the first electron, the second electron has to be removed from the following electronic configurations:
N:1s22s22p2
An atom having a more stable electronic configuration (half filled in case of N: 1s22s22p3) has a reduced tendency to lose electrons and consequently has a higher ionization energy.
C:1s22s22p1(removal of an electron from the 2p orbital will lead to a stable electronic configuration)
Be:1s22s1(removal of an electron from the 2s orbital will lead to a stable electronic configuration)
B:1s22s2(removing an electron from a fully-filled stable electronic configuration is difficult, leading to a higher ionisation energy)