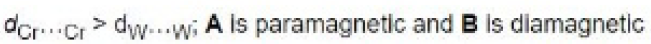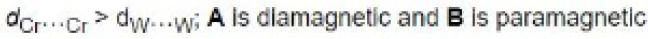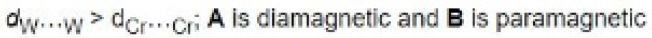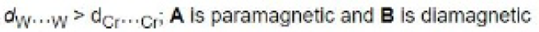Correct option is A
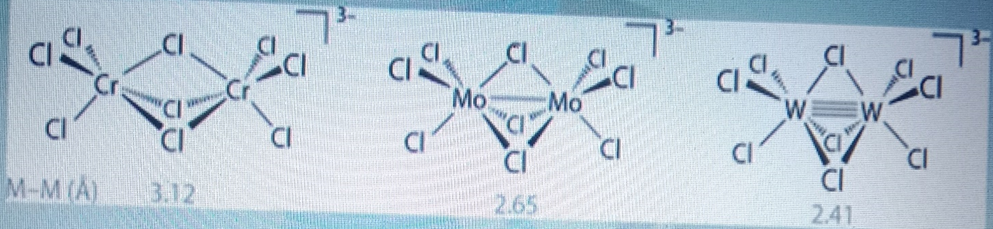
For [Cr2Cl9]3-, two CrCl3 fragments are held together by the three bridging chlorides. There is no direct Cr-Cr bond and hence it is paramagnetic with 6 unpaired electrons. For tungsten, there is good overlap (due to large orbitals) and a triple W-W bond with no unpaired electrons.