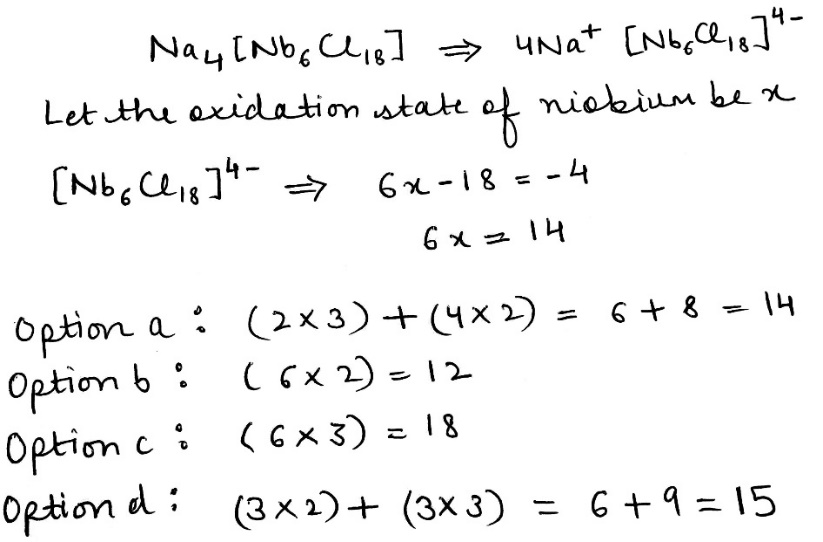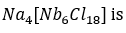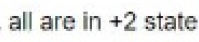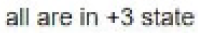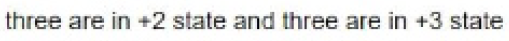Correct option is A
Metal cluster compounds are a molecular ion or neutral compound composed of three or more metals and featuring significant metal-metal interactions.
The halides of low-valent early metals often are clusters with extensive M-M bonding. The situation contrasts with the higher halides of these metals and virtually all halides of the late transition metals, where metal-halide bonding is replete.
Transition metal halide clusters are prevalent for the heavier metals: Zr, Hf, Nb, Ta, Mo, W, and Re.